Giant cell arteritis (GCA), the most common primary vasculitis in adults, is a granulomatous systemic vasculitis that affects the aorta and its major branches [1]. For decades, glucocorticosteroids (GCs) have served as the fundamental treatment for GCA, effectively reducing the risk of irreversible vision loss. However, disease control frequently requires prolonged GC exposure in older individuals, who often have comorbidities such as hypertension, diabetes, osteoporosis, cardiovascular disease, and sarcopenia [2]. Relapse during GC tapering remains the general rule rather than the exception, since more than 50% of patients experience GCA recurrence, which results in repeated exposure to rescue treatment courses and overall high cumulative toxicity [3].
Since the landmark GiACTA (Giant-Cell Arteritis Actemra) trial of tocilizumab (TCZ), a monoclonal antibody against the interleukin-6 (IL-6) receptor, focus has shifted to GC-sparing strategies [4]. Tocilizumab became the first approved GC-sparing treatment for GCA and contributed to reframing treatment goals to sustain remission while minimizing GC burden. Despite its efficacy, TCZ has limitations that are clinically relevant in routine practice. In a retrospective cohort, relapse was uncommon during TCZ therapy, but increased substantially after discontinuation, while adverse events – such as severe infections and diverticulitis – were also reported [5]. Furthermore, IL-6 blockade is well recognized for its ability to suppress acute-phase reactants such as C-reactive protein (CRP), thereby potentially limiting the detection of concurrent infections. In addition, a proportion of patients have contraindications to TCZ treatment, experience adverse events, or relapse [6, 7]. Collectively, these limitations justify the evaluation of alternative GC-sparing agents.
Mechanistically, the role of IL-6 and interferon-γ in GCA pathophysiology is well documented, with both signaling through the JAK-STAT (Janus kinase-signal transducer and activator of transcription) pathway (Fig. 1) [8, 9]. With this in mind, upadacitinib (UPA), an oral selective JAK1 inhibitor, represents a targeted therapeutic candidate for GC-sparing disease control. The SELECT-GCA trial, a multicenter randomized placebo-controlled study, recently confirmed the efficacy of UPA in patients with GCA [10]. The trial included patients over 50 years old, with a diagnosis of new-onset, or relapsing GCA, confirmed by imaging or biopsy. Participants were randomized to UPA 15 mg/day, UPA 7.5 mg/day, or placebo, combined with standardized GCs therapy. In the UPA arms, GCs were tapered over 26 weeks, while in the placebo arm, GCs were tapered over 52 weeks. The primary endpoints were sustained remission, defined as the absence of signs and symptoms of GCA from week 12 until week 52, and adherence to the protocol-specified GC tapering. Secondary outcomes included complete remission, disease flare-related endpoints, patient-reported outcome measures (PROMs), and cumulative exposure to GCs. The trial included 428 patients (70% newly diagnosed GCA), and at week 52 a total of 299 patients (69.9%) had completed the regimen. In the SELECT-GCA trial, UPA 15 mg administered with a 26-week GCs taper was superior to placebo plus a 52-week taper regarding sustained remission at week 52 (46.4% vs. 29.0%, p = 0.002). Furthermore, the UPA 15 mg arm received significantly less GCs. On the other hand, the UPA dose at 7.5 mg did not differ from placebo in sustaining remission (41.1% vs. 29.0%, respectively). It is important to note, however, that the definition of remission in GCA is not unanimous in trials, making interpretation of results and the incidence of adverse events difficult to compare between trials [11, 12].
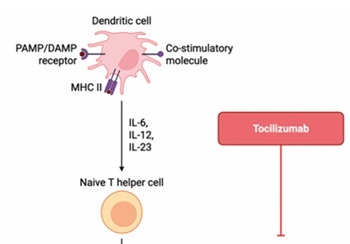
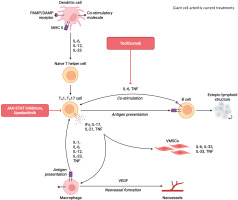
Fig. 1
Current treatments for giant cell arteritis (created with BioRender.com; #Bogdanos, D. (2026) https://BioRender.com/phvjqjj).
DAMP – damage-associated molecular pattern, IFγ – interferon γ, IL– interleukin, JAK-STAT – Janus kinase-signal transducer and activator of transcription, MHC – major histocompatibility complex, PAMP – pathogen-associated molecular pattern, TNF – tumor necrosis factor, VEGF – vascular-endothelial growth factor.
Regarding safety, it is known that older patients often carry baseline cardiovascular disease risk, while clinicians have also internalized regulatory warnings derived from rheumatoid arthritis populations regarding JAK inhibitors and major adverse cardiovascular events or malignancy [13]. It is important to note that in the SELECT-GCA trial [10], no major adverse cardiovascular events were recorded in the UPA-receiving arms. Safety outcomes were generally similar across groups. The incidence of adverse events leading to treatment discontinuation was lower with UPA than with placebo, while similarly, the incidence of serious infections was also lower with UPA compared to the placebo arm. With respect to adverse events of special interest, patients receiving UPA at a dose of 15 mg experienced herpes zoster infections more frequently, consistent with the JAK inhibitor class. This is encouraging but should be interpreted cautiously. A one-year event-driven comparison in a few hundred patients cannot definitively assess long-term cardiovascular or malignancy risk, and the trial authors themselves highlight the need for longer follow-up. Therefore, extension data will be pivotal for determining safety in patients with GCA.
Taken together, these findings position UPA as a potential additional GC-sparing option in GCA. However, several clinically relevant uncertainties remain. First, the efficacy of UPA after failure of TCZ is unknown [10], and future studies should incorporate TCZ-refractory populations, as this clinically important subgroup has not been represented in randomized trials to date. Second, comparative effectiveness remains undefined; randomized head-to-head trials with prespecified non- inferiority hypotheses are required. Third, incorporation of imaging-based endpoints alongside clinical remission, as well as evaluation of treatment duration and withdrawal strategies, is required to quantify the risk of relapse and define optimal treatment algorithms. Finally, given the profile of patients with GCA, longer-term follow-up is essential to define cardiovascular and malignancy risk in patients treated with UPA.
Overall, the SELECT-GCA trial supports UPA as an ef- fective GC-sparing option in GCA. However, further comparative studies are needed to clarify its optimal role in daily clinical practice.