Introduction
Fibromyalgia syndrome
Fibromyalgia syndrome (FMS) is a chronic disorder characterized by widespread musculoskeletal pain accompanied by a constellation of symptoms, notably fatigue, sleep and mood disturbances, and cognitive dysfunction [1]. It represents a significant clinical challenge, affecting approximately 2–8% of the general population [2, 3]. Fibromyalgia syndrome occurs across all global populations and ethnicities, predominantly affects women, and can manifest at any age, most commonly in early to middle adulthood [2–4].
Historically, the 1990 classification criteria of the American College of Rheumatology (ACR) required both chronic widespread pain and the presence of tenderness at ≥ 11 of 18 specific tender points upon physical examination. Updated ACR criteria in 2010 shifted toward a symptom-based approach, eliminating the tender point examination and instead utilizing the Widespread Pain Index (WPI) and Symptom Severity (SS) scale. The diagnosis was established when patient met either WPI ≥ 7 with SS ≥ 5 or WPI 3–6 with SS ≥ 9, symptoms were present for at least 3 months, and there was no other explanation of the pain. In 2016 a new criterion for generalized pain, defined as pain in at least 4 of 5 specific regions, was introduced. Additionally, the previous criterion of WPI ≥ 3 was modified to WPI ≥ 4, the recommendation to exclude patients with other diagnoses was removed, and the fibromyalgia severity scale (sum of the WPI and SS) was added [5, 6].
Despite growing evidence, the pathogenesis of FMS remains incompletely understood. A prevailing model implicates altered pain processing, characterized by central sensitization, a process of amplified neuronal signaling within the central nervous system (CNS) [7–9]. Patients with fibromyalgia experience an amplified perception of pain, which manifests as hyperalgesia (increased sensitivity to painful stimuli) and allodynia (pain resulting from stimuli not normally painful). The pathophysiology may be associated with dysfunctions in mono-aminergic neurotransmission, notably increased concentration of excitatory neurotransmitters such as glutamate and substance P, alongside reduced serotonin and norepinephrine levels within descending inhibitory pathways of the CNS. Dopaminergic dysregulation and altered endogenous opioid activity may also play a role [9]. Neuroimaging studies support these findings, showing abnormal activation patterns in pain-related brain regions and decreased functional connectivity in the descending pain-modulating system [10, 11]. Peripheral nerve abnormalities such as decreased epidermal nerve fiber density and increased spontaneous activity of mechanically insensitive C-fibers also play a crucial role. Inflammatory processes have been increasingly recognized in FMS pathophysiology, demonstrated by elevated cytokine levels, e.g., interleukin (IL)-6, IL-8, and immune cell activation [9, 12]. Genetic predisposition plays a substantial role in the development of FMS, with specific gene polymorphisms (e.g., SLC6A4, COMT) associated with altered pain perception and neurotransmitter metabolism [13, 14]. Other aspects considered are endocrine dysregulation, and psychosocial factors such as depression, anxiety, stress, and poor sleep quality [7, 9].
Small fiber neuropathy
Whereas FMS has unclear etiology, small fiber neuropathy (SFN) represents a well-characterized disorder with established pathogenesis and clearly identifiable underlying causes. It predominantly affects thinly myelinated Aδ and unmyelinated C fibers, which are responsible for thermal sensation and nociception, and autonomic function [15, 16]. Clinically, SFN presents with paresthesia, allodynia, hyperesthesia, and numbness, often in a distal and length-dependent distribution, and may be accompanied by autonomic manifestations including altered sweating, dry eyes or mouth, and erectile dysfunction in males. The most common recognized etiology is diabetes mellitus; however, other causes include impaired glucose tolerance, autoimmune diseases, hypothyroidism, amyloidosis, vitamin deficiency (mainly B12), infections, toxic exposures (alcohol), and hereditary sensory-autonomic neuropathies. Nevertheless, 30–50% of cases are idiopathic [15–17]. Diagnostic confirmation of SFN is challenging due to the normal findings in conventional nerve conduction studies, which assess only large fiber integrity. Therefore, diagnosis relies on tests, such as quantitative sensory testing (QST), quantitative sudomotor axon reflex testing (QSART), laser evoked potentials (LEP), autonomic testing, corneal confocal microscopy and skin biopsy with assessment of intraepidermal nerve fiber density (IENFD) [15–17]. The diagnostic criteria of SFN are: 1) clinical signs of small fiber impairment, 2) abnormal thermal detection thresholds on foot assessed by QST, and 3) reduced IENFD in skin biopsy, where 2 out of 3 must be met [16].
Skin biopsy
Skin biopsy with quantification of IENFD has become an important diagnostic tool in the evaluation of SFN [16, 18]. The method was originally developed at the Karolinska Institute and later standardized in centers such as the University of Minnesota and Johns Hopkins University. The standard technique involves a 3-mm punch biopsy from the distal leg – typically 10 cm above the lateral malleolus and, optionally, from the proximal thigh [18]. The procedure is simple, minimally invasive, well tolerated, and does not require suturing. Following the biopsy, the tissue is immediately fixed in cold fixative, then vertically sectioned and immunostained with antibodies against the protein gene product 9.5 (PGP 9.5). This allows for direct visualization and quantification of unmyelinated C-fibers crossing the dermalepidermal junction. Bright-field immunohistochemistry and immunofluorescence, with or without the use of confocal microscopy, are established techniques for identification of intraepidermal nerve fibers. A diagnosis of SFN is supported when IENFD falls below the 5th percentile of age- and sex-matched normative reference values [16, 18].
The first skin biopsy studies in FMS were conducted in the mid-1980s and used immunofluorescence techniques, revealing deposits of immunoglobulins, particularly immunoglobulin G, at the dermal–epidermal junction [19]. These early findings raised the possibility of an underlying immune-mediated or inflammatory mechanism contributing to fibromyalgia symptoms. In subsequent years, research focused on mast cells, collagen deposition, and signs of neurogenic inflammation, suggesting interactions between immune and neural pathways [20, 21]. Given that a subset of patients with fibromyalgia report neuropathic symptoms resembling those observed in SFN, researchers began investigating peripheral nervous system dysfunction as a potential underlying mechanism – particularly through the use of skin biopsy and assessment of IENFD. However, the explicit application of IENFD evaluation in fibromyalgia research emerged only in the early 21st century, marking a significant shift toward exploring structural peripheral abnormalities in this condition [22, 23].
The purpose of this systematic review was to summarize existing evidence on the use of skin biopsy in patients with fibromyalgia, with particular focus on the evaluation of IENFD as a marker of small fiber pathology (SFP).
Material and methods
This review was conducted in accordance with PRISMA guidelines. A literature search was performed using PubMed, Scopus, and Web of Science databases, as well as in other sources including the European Alliance of Associations for Rheumatology (EULAR) Abstract Archive, using the search terms “fibromyalgia,” “skin biopsy,” and “small fiber neuropathy.” The search included studies published up to December 2024.
Inclusion criteria were: availability of full text, original research articles, application of skin biopsy as a diagnostic method, and assessment of IENFD. Exclusion criteria comprised studies not published in English, animal studies, studies focused on conditions other than fibromyalgia, and studies involving less than 10 patients.
Results
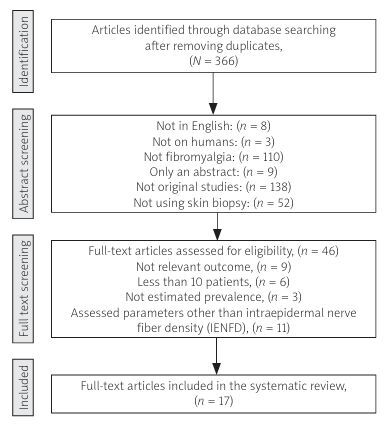
A total of 751 records were identified through database searches, and an additional 3 records were retrieved from EULAR abstracts. After abstract screening, 46 full-text articles were assessed for eligibility. Overall, 17 studies were included in the review. The article selection process is illustrated in the PRISMA flow diagram (Fig. 1).
The included studies were conducted globally, with 3 originating from the United States [22, 24, 25] and 14 from Europe, predominantly Italy and Germany [23, 25–37]. All studies applied the ACR 1990 or 2010 criteria as the standard for fibromyalgia classification. One of the included studies was conducted specifically in adolescents [25], whereas all other studies involved adult patients.
In most studies, skin biopsy was performed at the distal leg [22, 25, 28, 32, 35] or at both the distal leg and thigh [23, 24, 26, 29–31, 33, 34, 36–38], with 1 study also including the fingertip as a biopsy site [27]. Protein gene product 9.5 immunostaining was employed in all studies to quantify intraepidermal nerve fibers. Some researchers also used additional immunostaining methods such as staining with antibodies against growth-associated protein 43 to visualize regenerating nerve fibers [23], or with antibodies against calcitonin gene-related peptide, substance P, CD31, and neurofilament 200 for small fiber subtypes [32]. In addition to skin biopsy, several studies have employed other diagnostic methods such as QST, QSART, pain-related evoked potentials, LEP or corneal confocal microscopy [23, 24, 27, 29–31, 33, 35–37].
Consistent evidence of SFP in patients with fibromyalgia was demonstrated across all skin biopsy studies, which reported decreased IENFD compared to healthy controls. The estimated frequency of IENFD was reported across all studies, with prevalence ranging from approximately 30% to 85% [22–38]. The pattern of nerve fiber loss presented in some studies was heterogeneous. A length-dependent pattern – with greater IENFD loss in distal sites (e.g., lower leg) – appears in a minority of cases [30]. Non-length-dependent patterns are more prevalent, with reduced IENFD observed in 85% of FMS patients at the thigh, compared to approximately 12% at the distal leg [33]. Additionally, about 15% of patients present a generalized nerve fiber loss pattern, with significant reductions observed at both distal and proximal sites [30].
Table I contains a summary of all studies included in this review.
Table I
Data from all included studies
| Author | Year | Country | Sample size | Study group | Group size | Mean age | Sex (female/male) | Biopsy site | Prevalence number | Prevalence estimate (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Üçeyler et al. [23] | 2013 | Germany | 155 | Fibromyalgia Depression without pain Control | 24 10 121 | 59 (50–70) 50 (39–75) Unavailable | 22/2 9/1 Unavailable | Thigh and distal leg | 10 | 42 |
| Oaklander et al. [22] | 2013 | USA | 57 | Fibromyalgia Control | 27 30 | 47 (26–68) 45 (25–65) | 20/7 24/6 | Distal leg | 11 | 41 |
| Giannoccaro et al. [26] | 2013 | Italy | 52 | Fibromyalgia Control | 20 32 | 40 ±6 Unavailable | 19/1 Unavailable | Thigh and distal leg | 6 | 30 |
| De Tommaso et al. [27] | 2014 | Italy | 81 | Fibromyalgia Control | 21 60 | 51 ±9 53 ±6 | 18/3 50/10 | Thigh, distal leg, fingertip | 16 | 76 |
| Kosmidis et al. [28] | 2014 | Greece | 80 | Fibromyalgia Control | 46 34 | 53 (29–76) 32 (19–84) | 41/5 18/16 | Distal leg | 16 | 34 |
| Leinders et al. [29] | 2016 | Germany | 116 | Fibromyalgia Control | 28 88 | 51 (39–74) 44 (16–79) | 26/2 80/8 | Thigh or distal leg | 14 | 50 |
| Lawson et al. [24] | 2018 | USA | 155 | Fibromyalgia | 155 | 49 (18–87) | 105/50 | Thigh and distal leg | 62 | 40 |
| Evdokimov et al. [30] | 2019 | Germany | 248 | Fibromyalgia Depression with pain Control | 117 11 120 | 52 (22–75) 52 (43–58) 50 (20–84) | 117/0 11/0 120/0 | Thigh and distal leg | 74 | 63 |
| Fasolino et al. [31] | 2020 | Italy | 57 | Fibromyalgia | 57 | 46 ±10 | 54/3 | Thigh and distal leg | 18 | 32 |
| Evdokimov et al. [32] | 2020 | Germany | 192 | Fibromyalgia Control | 86 106 | 51 (23–74) 50 (20–84) | 86/0 106/0 | Distal leg | 38 | 44 |
| Vecchio et al. [33] | 2020 | Italy | 81 | Fibromyalgia | 81 | 50 ±10 | 73/7 | Thigh and distal leg | 69 | 85 |
| Boneparth et al. [25] | 2021 | USA | 38 | Fibromyalgia Control | 15 23 | 17 (16–18) 18 (16–19) | 14/1 21/2 | Distal leg | 8 | 53 |
| Quitadamo et al. [34] | 2023 | Italy | 62 | Fibromyalgia | 62 | 51 (26–71) | 58/4 | Thigh and distal leg | 52 | 84 |
| Leone et al. [35] | 2023 | Italy | 94 | Fibromyalgia Small fiber neuropathy | 64 30 | 47 ±11 54 ±14 | 60/4 23/7 | Distal leg | 20 | 31 |
| Jänsch et al. [36] | 2024 | Germany | 211 | Fibromyalgia Small fiber neuropathy | 158 53 | 52 (22–75) 53 (22–73) | 158/0 53/0 | Thigh and distal leg | 104 | 66 |
| Falco et al. [38] | 2024 | Italy | 138 | Fibromyalgia Small fiber neuropathy Control | 58 32 48 | 50 ±10 64 ±12 47 ±15 | 52/6 Unavailable Unavailable | Thigh and distal leg | 23 | 40 |
| Feulner et al. [37] | 2024 | Germany | 59 | Fibromyalgia Control | 42 17 | 56 (31–75) Unavailable | 0/42 0/17 | Thigh and distal leg | 35 | 83 |
Discussion
It is important to recognize that a considerable subset of patients with FMS may also exhibit impairment of peripheral small fibers. This line of research has initiated a new direction in the study of FMS, contributing to its recognition as a legitimate clinical condition and supporting the growing body of evidence indicating a peripheral component in the pathogenesis. Multiple independent case-control studies from different regions have consistently found lower IENFD in FMS patients compared to healthy controls. This observation extends beyond adult populations; Boneparth et al. [25] evaluated IENFD in adolescents with juvenile fibromyalgia (JFM) and found significantly reduced IENFD in 53% of JFM patients, compared to only 4% of age-matched healthy controls.
Notably, a study by Evdokimov et al. [30] provided an in-depth assessment of SFP in patients with FMS, identifying distinct patterns of nerve loss – distal, proximal, and generalized. Among these, patients with generalized IENFD reduction exhibited a more severe clinical profile, characterized by higher pain intensity, and greater functional impairment compared to those with preserved innervation [30]. In line with these findings, Quitadamo et al. [34] conducted an observational study assessing changes in IENFD over an 18-month period and similarly found that patients with reduced IENFD at both distal and proximal sites experienced more severe symptoms and poorer treatment outcomes. An additional noteworthy discovery from this study was that the decrease in IENFD did not progress over time [34]. These findings suggest a link between SFP and symptom severity in FMS; however, the underlying cause remains unclear.
Leinders et al. [29] investigated the relationship between miRNA, which is responsible for regulation of molecular factors controlling nerve regeneration, and SFP in patients with FMS. Aberrant expression of microRNAs in white blood cells of FMS patients was found, of which particularly miR-let-7d correlated with reduced IENFD. In addition, skin biopsies from FMS patients with decreased IENFD revealed aberrant expression of miR-let-7d and insulin-like growth factor-1 receptor. The exact pathophysiological significance of these findings remains to be further clarified; however, the authors suggest that it may be the missing link in pathogenesis of small fiber loss in FMS.
The study by Evdokimov et al. [30] aimed to characterize dermal skin innervation in addition to epidermal innervation. The results revealed a significant reduction in the length of nerve fibers associated with blood vessels in FMS patients compared to healthy controls, while the overall dermal nerve fiber length immunoreacted with PGP 9.5 did not differ. The authors speculate that it could explain symptoms such as impaired thermal tolerance reported in FMS patients, highlighting a possible vascular component in the pathology [32]. In contrast, another study, which evaluated cutaneous arteriole-venule shunt (AVS) innervation from the hypothenar region of FMS patients, found significantly increased peptidergic sensory nerve fibers within AVS in FMS patients. The author suggested that excessive innervation of AVS dysregulates blood flow, resulting in widespread pain in FMS [39]. The studies differed in biopsy site (hand vs. lower leg), which may have contributed to the observed discrepancies; therefore, further comparative research is needed.
Another important finding comes from a recent study by Feulner et al. [37], which focused on assessing SFP in an exclusively male cohort with FMS, addressing a gap in existing research predominantly centered on female populations. Skin biopsies from the lower leg and upper thigh revealed that 83% of participants exhibited reduced IENFD in at least 1 biopsy site. Notably, > 50% of male patients had generalized IENFD reduction, while proximal-only reduction was rare. This pattern differs from findings in women, warranting further sex-specific research.
Greater clarity is needed in distinguishing between SFN and FMS, particularly when interpreting skin biopsy results and considering diagnostic implications. Isolated decreased IENFD does not necessarily indicate the presence of a coexisting SFN or that SFN is the underlying cause of fibromyalgia symptoms. The recent study showed that FMS and SFN are clinically different conditions [36]. Jänsch et al. [36] directly compared 158 women with FMS and 53 with SFN. The study found that FMS and SFN are distinct clinical entities. FMS patients typically experienced symptom onset at a younger age (~10 years), reported more intense and widespread pain, often presented with comorbidities such as irritable bowel syndrome and sleep disturbances, and family history was positive for chronic pain and mood disorders. In contrast, SFN patients more commonly reported localized neuropathic symptoms such as paresthesia and numbness, frequently associated with impaired glucose metabolism and a familial background of neurological diseases. Moreover, although both groups showed reduced IENFD, the pattern of fiber loss differed. Skin biopsies revealed reduced IENFD in 66.2% of FMS patients, predominantly at proximal sites, contrasting with more distal denervation in SFN. These findings emphasize that decreased IENFD should not be interpreted in isolation but rather in conjunction with the patient’s symptom profile, history, and physical examination findings.
Beyond the well-documented reduction in IENFD in FMS, several isolated studies have noted other skin biopsy abnormalities [39–45]. One study found an increase in mast cells in the papillary dermis of FMS patients (5–14 mast cells per field) compared to controls (0–1 mast cells, p < 0.001), strongly positive for α1-antitrypsin, and overexpression of proteinase-activated receptor 2; in contrast, expression of monocyte chemoattractant protein-1 and vascular endothelial growth factor was significantly lower in FMS [40]. A small study demonstrated up-regulation of δ- and κ-opioid receptors in the skin of FMS patients, as a first study investigating those receptors outside CNS [41]. The expression of pro-inflammatory cytokines (IL-1β, IL-6, IL-8, tumor necrosis factor α) in the skin was another line of research, which yielded conflicting results [42, 43]. Additionally, electron microscope imaging noted unusual unmyelinated fiber morphology – such as “ballooned” Schwann cell sheaths around C-fibers – in a subset of fibromyalgia cases compared to controls [44]. The study by Sánchez-Domínguez et al. [45] demonstrated oxidative stress and mitochondrial dysfunction in skin biopsies from FMS patients. The above findings are rather preliminary and have not yet been widely replicated, so they should be interpreted with caution; nonetheless, they suggest potential peripheral contributors to fibromyalgia pain that warrant further investigation.
The potential therapeutic implications of skin biopsy findings in FMS remain an open question. The presence of SFP, suggesting a neuropathic pain component, may lead to the hypothesis that patients with SFP could respond more favorably to neuropathic pain medications such as pregabalin or duloxetine, both of which are approved for the treatment of FMS. However, no prospective studies have yet confirmed this, and evidence supporting differential treatment strategies remains lacking. A pilot study by Metyas et al. [46] reported that intravenous immunoglobulin treatment led to improvements in nerve fiber density in a small group of patients with coexisting FMS and SFN. However, this is currently the only study of its kind, with a very limited sample size. A surprising finding was reported in a supervised, home-based multicomponent physical activity program implemented over 18 months in FMS patients: IENFD significantly increased at both proximal and distal skin biopsy sites [47]. Remarkably, this regeneration of small fibers correlated with clinical improvement, as measured by reductions in Fibromyalgia Impact Questionnaire scores.
Conclusions
Small fiber pathology is present in a significant proportion of patients with FMS. Skin biopsy represents a simple and valuable tool for detecting this pathology and may assist in identifying distinct clinical subgroup of FMS individuals. Further research into the underlying mechanisms of SFP in FMS is needed to clarify its pathophysiological role and to support the integration of skin biopsy into routine diagnostic evaluation of FMS.