Introduction
Capillaroscopy is a non-invasive method that involves imaging the capillary vessels of the verrucous layer of the skin within the fingernail fold with a stereoscopic microscope or video capillaroscope. This examination is used in the differential diagnosis between primary Raynaud’s phenomenon (pRP) and microcirculation disorders in secondary Raynaud’s phenomenon (sRP), accompanying systemic connective tissue diseases [1, 2]. Capillaroscopy is of particular importance in the early diagnosis of systemic sclerosis (SSc). In addition, this examination is also used in the assessment of disease activity and the degree of involvement of internal organs, with a few reports indicating the use of capillaroscopy in monitoring and assessing the effectiveness of the therapy applied. Furthermore, recent studies highlight the importance of capillaroscopy in differentiating the severity of vascular damage in limited and diffuse cutaneous SSc [3].
In the course of SSc, characteristic microcirculation disorders are present in the capillaroscopic examination, referred to as microangiopathy typical of SSc, i.e. “scleroderma pattern.” The term scleroderma microangiopathy includes the presence of dilated vessels with a diameter of more than 50 μm, referred to as giant capillaries, or a significant reduction in capillary density, with the presence of loops with abnormal morphology and, in particular, neoangiogenesis. Moreover, microhaemorrhages are an additional element of scleroderma microangiopathy [4, 5]. Capillaroscopic changes that do not meet the criteria of scleroderma microangiopathy are referred to as non-scleroderma patterns and include normal images and non-specific abnormalities (Table I) [1, 6]. The vast majority of non-specific abnormalities that may occur in the healthy population may make it difficult for an inexperienced capillaroscopist to classify an image as a “scleroderma pattern” or a “non-scleroderma pattern.” To address this issue, the European Alliance of Associations for Rheumatology (EULAR) research group developed the “Fast Track” algorithm, which, after validation, provided a fast and simple, high-reliability image classification [7]. The “Fast Track” algorithm consists of 3 simple rules: Rule 1: Normal capillary density (≥ 7 capillaries) and the absence of giant capillaries allow the assessor to classify the capillaroscopic image as a non-scleroderma pattern. Rule 2: Extremely reduced capillary density (≤ 3 capillaries) together with abnormal shapes (i.e., “late scleroderma pattern”), or the presence of giant capillaries allow the assessor to classify the capillaroscopic image as a “scleroderma pattern.” Rule 3: If the imaging findings do not meet either Rule 1 or Rule 2, then the image is automatically scored as a “non-scleroderma pattern” [1, 7].
Table I
Division of capillaroscopic changes: scleroderma and non-scleroderma patterns [6]
Capillaroscopic examination in polymyositis and “scleroderma spectrum” diseases
In addition to SSc, capillaroscopic examination is helpful in the diagnosis and monitoring of “scleroderma spectrum” diseases, which include dermatomyositis (DM), mixed connective tissue disease (MCTD), or undifferentiated connective tissue disease (UCTD) [8]. In systemic connective tissue diseases other than SSc, particularly in “scleroderma spectrum” diseases, capillaroscopic images may present normal capillaroscopic findings, nonspecific abnormalities, or “scleroderma patterns” [1]. According to the recommendations of Cutolo et al. [5], capillaroscopic changes characteristic of SSc that meet the criteria of the “scleroderma pattern” occurring in other diseases of the “scleroderma spectrum” are referred to as scleroderma-like microangiopathy. Capillaroscopic examination has been shown to be a valuable tool for assessing microcirculation disorders in idiopathic inflammatory myopathies (IIMs), especially in those subtypes in which vascular damage plays a significant role, namely in DM, or anti-synthetase syndrome [9]. Scleroderma-like microangiopathy occurs in about 20–40% of patients with IIMs, more often in DM than in polymyositis [10]. The capillaroscopic changes that dominate in the course of DM are increased neoangiogenesis and reduction in capillary density, with a relatively small number of avascular areas. The presence of branching giant vessels is also characteristic (Fig. 1). Often, in 1 patient, different capillaroscopic changes are found in different fingers [10]. Due to significant differences in capillaroscopic images in patients with SSc, compared to DM patients, some researchers suggest that the term “microangiopathy in the course of DM” should be used instead of “scleroderma-like microangiopathy” [11]. Many studies have emphasized that capillaroscopic images in the course of DM are very diverse [11, 12]. The analysis of 2 comparative studies on patients with SSc and DM shows that patients with DM seem to demonstrate less capillary loss than patients with SSc. In the scleroderma group, progression of microangiopathy from “early” to “late” stages over time is typical, which is not as pronounced in patients with DM [11, 12]. Specifically, comparing patients with DM with SSc patients with “early”, “active”, or “late” microangiopathy, DM patients generally show a smaller reduction in capillary density than patients with SSc, although they still show greater reduction than patients with early scleroderma microangiopathy in the course of SSc [11]. Interestingly, in the course of inflammatory myopathies, the “scleroderma-like” pattern, defined as a capillary pattern showing mixed microvascular markers of the scleroderma patterns, but not proportionally fitting the definition for the single “early,” “active,” or “late” pattern, was also found to be discriminatory between patients with IIMs and patients with SSc [13]. For example, in the course of inflammatory myopathies, a significant reduction of capillary density below 4 capillaries per linear millimeter may coexist with the presence of giant vessels, allowing such changes to be defined as a “scleroderma-like” pattern. In the course of microangiopathy in SSc, giant loops never co-occur with extreme reduction of the capillary density; namely, giant loops are characteristic of the “early scleroderma pattern,” whereas extreme vessel reduction without the presence of giant capillaries is characteristic of the “late scleroderma pattern” [1].
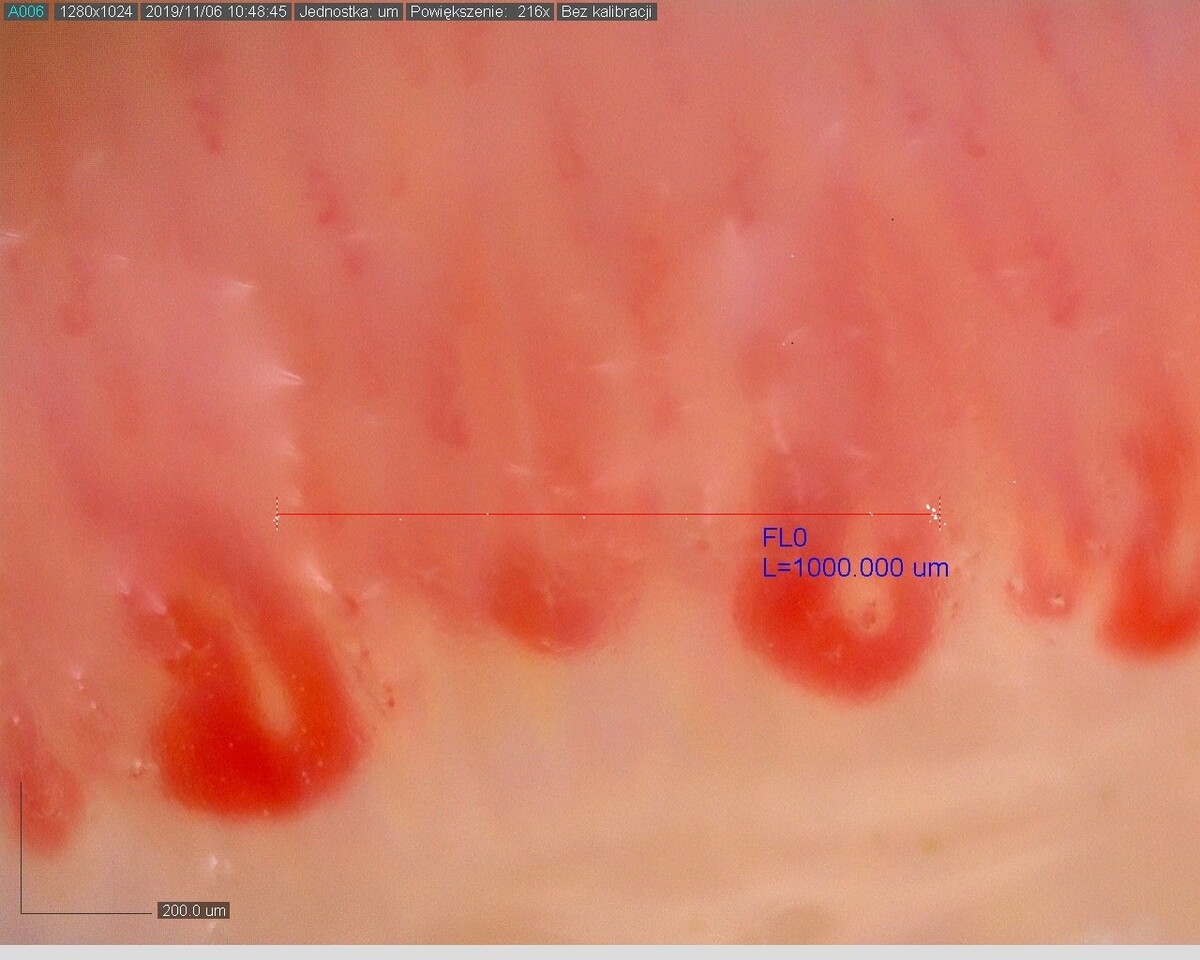
Fig. 1
Scleroderma-like microangiopathy in the course of dermatomyositis (200× magnification).
Source: Ewa Wielosz archive images.
Studies evaluating the relationship between microcirculation disorders in capillaroscopic examination of nailfold vessels and the activity and degree of internal organ involvement in the course of inflammatory myopathies are not unambiguous and require more research. In their study, Mugii et al. [14] demonstrated a significant relationship between capillaroscopic changes and the activity of DM. Significantly higher creatine kinase concentrations were found in the group of patients with scleroderma-like microangiopathy, compared to the group of patients without microangiopathy. Moreover, during the 9-month follow-up, capillaroscopic improvement was found (reduction in the number of giant capillaries and microhaemorrhages and increased capillary density), which correlated with clinical stabilization [14]. The authors of the study suggest that the improvement in microcirculation may be the result of immunosuppressive treatment used in the studied group of patients, which reflects the dynamics of capillaroscopic changes under the influence of therapy. In another study, Shenavandeh et al. [15] found a relationship between the overall assessment of DM activity on a visual analogue scale and a higher capillaroscopic index. Barth et al. [16], on the other hand, found a correlation between the number of vessels and lung involvement in patients with juvenile DM. In the group of patients with reduced capillary density, lower percentage values of forced vital capacity, total lung capacity, and carbon monoxide diffusing capacity were found, compared to patients with a normal number of vessels [16]. Thus, the assessment of capillary density is an important tool in monitoring inflammatory myopathies, especially juvenile DM, and rapid capillary loss is associated with their more severe course [17]. Therefore, in 2017, recommendations were developed including a list of tests that should be performed in patients with juvenile DM in order to monitor the disease and assess its activity and response to treatment. Among these tests, capillaroscopic examination of nailfold vessels is recommended [18].
Capillaroscopic patterns in MCTD are very diverse, ranging from normal patterns, through nonspecific changes, to scleroderma-like patterns. Capillaroscopic changes in the form of scleroderma-like microangiopathy are found in about 50–60% of patients with MCTD (Fig. 2) [19]. In 1 comparative study, which examined patients with MCTD, SSc, and systemic lupus erythematosus (SLE), the “scleroderma-like” pattern was detected in 64% of patients with MCTD [19]. Interestingly, in a large prospective observational study lasting 4.8 years in a cohort of 3,029 patients with RP, a significant association was observed between the presence of a “scleroderma-like” pattern and the development of MCTD [20]. There are studies that have shown an association of scleroderma-like microangiopathy, especially the presence of giant vessels, with interstitial lung disease (ILD) and pulmonary arterial hypertension in patients with MCTD, and reduced capillary density correlated with a more severe course of this disease [21, 22]. Nevertheless, Ornowska et al. [23] described 2 cases of patients with MCTD and ILD. Both of them had a decrease in capillary density in nailfold capillaroscopy. However, in the first patient, the capillaroscopic image was connected with severe lung disease with coexisting pulmonary arterial hypertension. In the second case, the course of ILD was mild, which indicates that the decrease in capillary number was irrelevant. Therefore, these data require confirmation in a larger number of patients [23]. Generally, patients with MCTD, compared to patients with SSc, are characterized by a smaller degree of reduction in capillary density and fewer avascular areas in capillaroscopic examination [24]. In 1 study, during a 3-year follow-up, a lesser progression of capillaroscopic changes in MCTD was demonstrated, compared to SSc, in the form of less vessel reduction and a smaller number of giant vessels [25]. In another comparative study evaluating capillaroscopic findings in patients with MCTD and those with UCTD, a significantly higher frequency of the “scleroderma-like” pattern was demonstrated in patients with MCTD [22]. Despite the occurrence of various capillaroscopic changes in MCTD, identification of a specific capillaroscopic pattern in the course of this disease is impossible due to the coexistence of similar changes in other scleroderma spectrum diseases. Diagnostic and prognostic value may only be significant when other clinical and serological aspects of the disease are taken into account [26, 27].
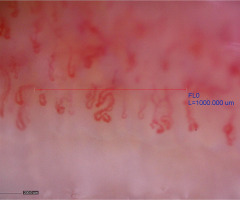
Fig. 2
Scleroderma-like microangiopathy in the course of mixed connective tissue disease (200× magnification).
Source: Ewa Wielosz archive images.
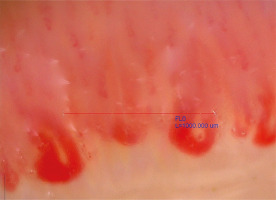
In the course of UCTS, most patients have normal capillaroscopic images. In about 40–50% of patients, nonspecific changes are found in the form of loops that cross several times, are deformed, dilated, but do not meet the definition of giant capillaries [28]. About 10% of patients meet the criteria of scleroderma-like microangiopathy, and this group in particular requires constant monitoring and observation for a specific systemic connective tissue disease, especially SSc [22]. In 1 study, Pizzorni et al. [22] performed a retrospective evaluation of capillaroscopic images in a group of patients with MCTD, UCTD, and pRP. Capillaroscopic changes of scleroderma-like microangiopathy type were found statistically significantly more often in the group of patients with MCTD, compared to UCTD. Moreover, the presence of neoangiogenesis, giant capillaries and capillary reduction was more marked in MCTD than in UCTD. The authors concluded that capillaroscopic changes in the course of UCTD resemble the picture found in pRP (nonspecific changes), in contrast to MCTD, where scleroderma-like microangiopathy and a reduction in the number of capillaries predominate [22].
Capillaroscopic examination in systemic lupus erythematosus
A meta-analysis of 40 studies conducted by the authors of the EULAR Study Group on Microcirculation in Rheumatic Diseases showed that in patients with SLE, dilated vessels, abnormal vascular morphology, and microbleeding were more frequently found in a capillaroscopic examination of nailfold vessels, compared to the healthy population [27]. Furthermore, “nonspecific” and “scleroderma-like” patterns were observed statistically significantly more often in SLE [29]. Most of the data on the relationship between capillaroscopic changes and organ symptoms in SLE require confirmation in a larger number of patients. Correlations between capillaroscopic changes and general disease activity in SLE have been described, and an association with abnormal vascular morphology and the presence of microhaemorrhages and SLE activity has been found [30]. Multicenter studies are still ongoing to clarify the relationship between capillaroscopic parameters and clinical and serological characteristics of SLE patients.
Capillaroscopic examination in Sjögren’s disease
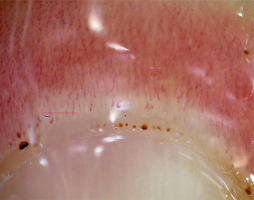
In the course of Sjögren’s disease (SjD), capillaroscopic findings are very variable, ranging from “normal” to “scleroderma-like” patterns. A meta-analysis of 7 observational studies of capillaroscopic changes in SjD showed that there is limited evidence of differences between patients and healthy volunteers [31]. One study found lower capillary density in SjD patients, compared to healthy volunteers, while another study found a higher frequency of capillaroscopic abnormalities in SjD patients with RP, compared to the group without RP [32, 33]. Most capillaroscopic patterns indicate “nonspecific” changes (Fig. 3). Capillaroscopic abnormalities of the “scleroderma-like” microangiopathy type are found in the subgroup of patients with the overlap syndrome of SjD/SSc or SjD/MCTD [31]. In summary, capillaroscopy in SjD is a particularly useful tool for patients with symptoms suggestive of the overlap syndrome – such as RP, digital swelling, or presence of antibodies other than marker antibodies for SjD – as it may help identify patients at risk of developing another systemic connective tissue disease, such as SSc or MCTD [31].
Capillaroscopic examination in antiphospholipid syndrome
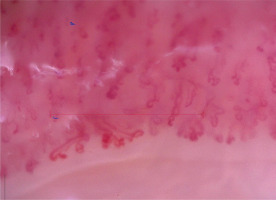
In primary antiphospholipid syndrome (APS), no specific capillaroscopic patterns are observed, but microcirculation abnormalities are often disclosed [34]. Observational studies have revealed “nonspecific” capillaroscopic patterns, such as dilated or abnormally shaped capillaries, suggesting microcirculation damage related to the presence of antiphospholipid antibodies [35]. The characteristic capillaroscopic pattern sometimes observed in patients with APS is referred to as the “comb” pattern and is characterized by parallel hemorrhages under the capillaries of the nailfold, mimicking the appearance of a comb (Fig. 4). Two recently published observational studies suggested that the “comb” pattern may serve as a potential capillaroscopic biomarker in patients with APS, as it is more frequently observed in this group of patients, compared to those with antiphospholipid antibodies not fulfilling the criteria for APS [36]. However, larger studies are needed to determine the usefulness of capillaroscopy in the diagnosis and monitoring of APS.
Insight into the future
Brzezińska et al. [37] conducted a pilot study aimed at evaluating nailfold capillaroscopy images using specialized software. The study yielded promising results. After analyzing 200 capillaroscopy images, the software demonstrated approximately 89% sensitivity and specificity. It enables rapid and precise assessment of capillary density per millimeter. The tool represents a viable alternative for clinical application. However, due to the lack of comprehensive capillary morphology analysis, further refinement is necessary.
Summary
Capillaroscopic examination of the nailfold vessels is used not only in the diagnosis and assessment of the activity of SSc, but also in sRP in polymyositis and “scleroderma spectrum” diseases or other systemic connective tissue diseases. However, prospective studies on larger groups of patients are required. Standardization of the description and interpretation of capillaroscopic examinations plays a key role in ongoing research, as well as in everyday medical practice. Further studies on standardized and long-term assessment of capillaroscopic changes may be of significant importance in making therapeutic decisions, changing treatment methods and monitoring patients with systemic connective tissue diseases.