Bieżący numer
Archiwum
Online first
O czasopiśmie
Redakcja
Rada Naukowa
Wydawca
Standardy etyczne i procedury
Bazy indeksacyjne
Recenzenci
Recenzenci Honorowi
Prenumerata
Kontakt
Najczęściej czytane artykuły
Dla autorów
Opłata za przetwarzanie artykułu (APC)
Książki i Konferencje
Książki
Konferencje
SARS CoV-2/COVID-19
PRACA ORYGINALNA
Neuropathic pain in rheumatoid arthritis: prevalence and associated factors
1
Department of Rheumatology, Charles Nicolle Hospital, Tunis, Tunisia
Data nadesłania: 30-03-2025
Data ostatniej rewizji: 04-05-2025
Data akceptacji: 04-08-2025
Data publikacji: 26-03-2026
Reumatologia 2026;64(2):132-138
SŁOWA KLUCZOWE
DZIEDZINY
STRESZCZENIE
Introduction:
This study aimed to assess the prevalence of neuropathic pain in rheumatoid arthritis (RA) patients, and to identify associated factors.
Material and methods:
We conducted a cross-sectional study in patients with RA. Neuropathic pain was evaluated using the DN4 (Douleur Neuropathique 4) and PainDETECT scores. Agreement between the 2 questionnaires was estimated through Cohen’s k. Factors associated with neuropathic pain were assessed.
Results:
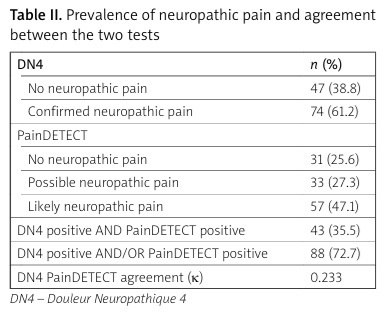
A total of 121 patients with RA were included, with a mean age of 57 ±12.2 years, and a predominance of women (83%). The mean duration of RA was 16 ±1.0 years. The mean Disease Activity Score in 28 joints (DAS28) was 4.28 ±1.0, indicating moderate activity of the RA. According to the DN4 score, 61.2% of patients had neuropathic pain, and according to the PainDETECT score, 47.1% of patients were identified as having possible or likely neuropathic pain. Eighty-four patients (69.4%) had a positive DN4 score and/or a positive PainDETECT score. The coefficient of agreement k between the 2 tests was 0.233, indicating a fair agreement. Factors significantly associated with the presence of neuropathic pain were: female sex (p = 0.01), prolonged morning stiffness duration (p = 0.01), number of painful joints (p = 0.01), and number of swollen joints (p = 0.03). Additionally, the use of glucocorticosteroids (p = 0.001), analgesics (p = 0.03) and anti-CD20 drugs (p = 0.03) were significantly associated with neuropathic.
Conclusions:
Our study suggests a potentially high prevalence of neuropathic pain in patients with RA, highlighting the need for early detection and appropriate management.
This study aimed to assess the prevalence of neuropathic pain in rheumatoid arthritis (RA) patients, and to identify associated factors.
Material and methods:
We conducted a cross-sectional study in patients with RA. Neuropathic pain was evaluated using the DN4 (Douleur Neuropathique 4) and PainDETECT scores. Agreement between the 2 questionnaires was estimated through Cohen’s k. Factors associated with neuropathic pain were assessed.
Results:
A total of 121 patients with RA were included, with a mean age of 57 ±12.2 years, and a predominance of women (83%). The mean duration of RA was 16 ±1.0 years. The mean Disease Activity Score in 28 joints (DAS28) was 4.28 ±1.0, indicating moderate activity of the RA. According to the DN4 score, 61.2% of patients had neuropathic pain, and according to the PainDETECT score, 47.1% of patients were identified as having possible or likely neuropathic pain. Eighty-four patients (69.4%) had a positive DN4 score and/or a positive PainDETECT score. The coefficient of agreement k between the 2 tests was 0.233, indicating a fair agreement. Factors significantly associated with the presence of neuropathic pain were: female sex (p = 0.01), prolonged morning stiffness duration (p = 0.01), number of painful joints (p = 0.01), and number of swollen joints (p = 0.03). Additionally, the use of glucocorticosteroids (p = 0.001), analgesics (p = 0.03) and anti-CD20 drugs (p = 0.03) were significantly associated with neuropathic.
Conclusions:
Our study suggests a potentially high prevalence of neuropathic pain in patients with RA, highlighting the need for early detection and appropriate management.
REFERENCJE (32)
1.
Englbrecht M, Tarner IH, van der Heijde DM, et al. Measuring pain and efficacy of pain treatment in inflammatory arthritis: a systematic literature review. J Rheumatol Suppl 2012; 90: 3–10, DOI: 10.3899/jrheum.120335.
2.
Raja SN, Carr DB, Cohen M, et al. The revised International Association for the Study of Pain definition of pain: concepts, challenges, and compromises. Pain 2020; 161: 1976–1982, DOI: 10.1097/j.pain.0000000000001939.
3.
Junker U. Chronic pain: the “mixed pain concept” as a new rational. Dtsch Arztebl Int 2004; 101: 1393.
4.
Bonezzi C, Fornasari D, Cricelli C, et al. Not all pain is created equal: basic definitions and diagnostic work-up. Pain Ther 2020; 9 (Suppl 1): 1–15, DOI: 10.1007/s40122-020-00217-w.
5.
Cohen SP, Vase L, Hooten WM. Chronic pain: an update on burden, best practices, and new advances. Lancet 2021; 397: 2082–2097, DOI: 10.1016/S0140-6736(21)00393-7.
6.
Moseley GL, Butler DS. Fifteen Years of Explaining Pain: The Past, Present, and Future. J Pain 2015; 16: 807–813, DOI: 10.1016/j.jpain.2015.05.005.
7.
Shraim MA, Massé-Alarie H, Hall LM, Hodges PW. Systematic Review and Synthesis of Mechanism-based Classification Systems for Pain Experienced in the Musculoskeletal System. Clin J Pain 2020; 36: 793–812, DOI: 10.1097/AJP.0000000000000860.
8.
Bailly F, Cantagrel A, Betin P, et al. Part of pain labelled neuropathic in rheumatic disease might be rather nociplastic. RMD Open 2020, 6: e001326, DOI: 10.1136/rmdopen-2020-001326.
9.
Minhas D, Clauw DJ. Pain Mechanisms in Patients with Rheumatic Diseases. Rheum Dis Clin North 2021; 47: 133–148, DOI: 10.1016/j.rdc.2021.01.001.
10.
Kentaro N, Tajima M, Oto Y, et al. How do neuropathic pain-like symptoms affect health-related quality of life among patients with rheumatoid arthritis?: A comparison of multiple pain- related parameters. Mod Rheumatol 2020; 30: 828–834, DOI: 10.1080/14397595.2019.1650462.
11.
Dubois-Mendes SM, Sa KN, Meneses FM, et al. Neuropathic pain in rheumatoid arthritis and its association with Afro-descendant ethnicity: a hierarchical analysis. Psychol Health Med 2021; 26: 278–288, DOI: 10.1080/13548506.2020.1749677.
12.
Freynhagen R, Baron R. The role of peripheral and central mechanisms in the development and persistence of neuropathic pain in rheumatoid arthritis. Rheumatology 2013; 52: 690–698.
13.
Aletaha D, Neogi T, Silman AJ, et al. Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 2010; 62: 2569–2581, DOI: 10.1002/art.27584.
14.
Tebaa H, Chekkouri EE, Arabi H, et al. La douleur neuropathique (DN) dans la spondyloarthrite et la polyarthrite rhumatoïde: prévalence, association avec les paramètres de la maladie, l’état fonctionnel et la qualité de vie: à propos de 181 cas. Rev Rhum 2022; 89 (Suppl 1): A125, DOI: 10.1016/j.rhum. 2022.10.180 (1).
15.
Freynhagen R, Baron R, Gockel U, Tölle TR. Pain DETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Curr Med Res Opin 2006; 22: 1911–1920, DOI: 10.1185/030079906X132488.
16.
Bouhassira D, Attal N, Alchaar H, et al. Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain 2005; 114: 29–36, DOI: 10.1016/j.pain.2004.12.010.
17.
Chatila N, Pereira B, Maarrawi J, Dallel R. Validation of a New Arabic Version of the Neuropathic Pain Diagnostic Questionnaire (DN4). Pain Pract 2017; 17: 78–87, DOI: 10.1111/papr.12419.
18.
de Araújo Pereira F, de Almeida Lourenço M, de Assis MR. Evaluation of peripheral neuropathy in lower limbs of patients with rheumatoid arthritis and its relation to fall risk. Adv Rheumatol 2022; 62: 9, DOI: 10.1186/s42358-022-00238-3.
19.
Martins Rocha T, Pimenta S, Bernardo A, et al. Determinants of non-nociceptive pain in Rheumatoid Arthritis. Acta Reumatol Port 2018; 43: 291–303.
20.
Baskozos G, Hébert HL, Mv Pascal M, et al. Epidemiology of neuropathic pain: an analysis of prevalence and associated factors in UK Biobank. Pain Rep 2023; 8: e1066, DOI: 10.1097/PR9.0000000000001066.
21.
Ahmed S, Magan T, Vargas M, et al. Use of the painDETECT tool in rheumatoid arthritis suggests neuropathic and sensitization components in pain reporting. J Pain Res 2014; 14: 579–588, DOI: 10.2147/JPR.S69011.
22.
McVinnie DS. Obesity and pain. Br J Pain 2013; 27: 163–170, DOI: 10.1177/2049463713484296.
23.
Filatova ES, Erdes SF. Etiopathogenetic factors of peripheral neuropathic pain in rheumatoid arthritis. Zh Nevrol Psikhiatr Im S S Korsakova 2017; 117: 67–71 [Article in Russian], DOI: 10.17116/jnevro20171178167-71.
24.
Lee YC, Cui J, Lu B, et al. Pain persists in DAS28 rheumatoid arthritis remission but not in ACR/EULAR remission: a longitudinal observational study. Arthritis Res Ther 2011; 13: R83.
25.
Hashizume H, Rutkowski MD, Weinstein JN, DeLeo JA. Central administration of methotrexate reduces mechanical allodynia in an animal model of radiculopathy/sciatica. Pain 2000; 87: 159–169, DOI: 10.1016/S0304-3959(00)00281-5.
26.
Rifbjerg-Madsen S, Christensen AW, Christensen R, et al. Pain and pain mechanisms in patients with inflammatory arthritis: A Danish nationwide cross-sectional DANBIO registry survey. PLoS One 2017; 12: e0180014, DOI: 10.1371/journal.pone. 0180014.
27.
Martin K, Bentaberry F, Dumoulin C, et al. Neuropathy associated with leflunomide: a case series. Ann Rheum Dis 2005; 64: 649–650, DOI: 10.1136/ard.2004.027193.
28.
Kho LK, Kermode AG. Leflunomide-induced peripheral neuropathy. J Clin Neurosci 2007; 14: 179–181, DOI: 10.1016/j.jocn. 2005.08.021.
29.
Tsouni P, Bill O, Truffert A, et al. Anti-TNF alpha medications and neuropathy. J Peripher Nerv Syst 2015; 20: 397–402, DOI: 10.1111/jns.12147.
30.
Maisons V, Kaysi S. Rituximab-associated neuropathy: About two cases. Eur J Intern Med 2020; 76: 102–103, DOI: 10.1016/ j.ejim.2020.03.011.
31.
Mavragani CP, Vlachoyiannopoulos PG, Kosmas N, et al. A case of reversible posterior leucoencephalopathy syndrome after rituximab infusion. Rheumatology (Oxford) 2004; 43: 1450–1451, DOI: 10.1093/rheumatology/keh305.
32.
Michael D, Weiss MD, Pamela Becker MD. Paradoxical worsening of anti-myelin-associated glycoprotein polyneuropathy following rituximab. Muscle Nerve 2014; 49: 457–458, DOI: 10.1002/mus.23989.
Copyright: © Narodowy Instytut Geriatrii, Reumatologii i Rehabilitacji w Warszawie. This is an Open Access journal, all articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0) License (https://creativecommons.org/licenses/by-nc-sa/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, provided the original work is properly cited and states its license.
Udostępnij
ARTYKUŁ POWIĄZANY
Przetwarzamy dane osobowe zbierane podczas odwiedzania serwisu. Realizacja funkcji pozyskiwania informacji o użytkownikach i ich zachowaniu odbywa się poprzez dobrowolnie wprowadzone w formularzach informacje oraz zapisywanie w urządzeniach końcowych plików cookies (tzw. ciasteczka). Dane, w tym pliki cookies, wykorzystywane są w celu realizacji usług, zapewnienia wygodnego korzystania ze strony oraz w celu monitorowania ruchu zgodnie z Polityką prywatności. Dane są także zbierane i przetwarzane przez narzędzie Google Analytics (więcej).
Możesz zmienić ustawienia cookies w swojej przeglądarce. Ograniczenie stosowania plików cookies w konfiguracji przeglądarki może wpłynąć na niektóre funkcjonalności dostępne na stronie.
Możesz zmienić ustawienia cookies w swojej przeglądarce. Ograniczenie stosowania plików cookies w konfiguracji przeglądarki może wpłynąć na niektóre funkcjonalności dostępne na stronie.