Introduction
The aim of this literature review is to challenge a common misconception that elevated erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels, halo sign on temporal artery ultrasound, and arteritic anterior ischemic optic neuropathy (A-AION) are essential to make a diagnosis of giant cell arteritis (GCA). The discussed issues are supplemented by the case description of a 70-year-old woman with a biopsy-proven GCA who met the 2022 American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (EULAR) Classification Criteria for Giant Cell Arteritis. The patient had left posterior ischemic optic neuropathy (PION), ESR and CRP levels were within normal limits, and she had no halo sign on the temporal artery ultrasound.
Material and methods
The literature review adhered to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 2020 guidelines as in the presented flow diagram (Fig. 1) to ensure transparency and rigor in the review process. A comprehensive literature search was conducted via PubMed, using the key words “giant cell arteritis”, “GCA”, “posterior ischemic optic neuropathy”, “PION”, “ESR”, “CRP”, “halo sign”, “temporal artery ultrasound”, “temporal artery biopsy”, and “A-AION” in various combinations. Original research, literature reviews, case reports, case series, and letters to the editors were included. Articles that were non-English language, irrelevant to the research questions, or outdated were excluded.
Case description
A 70-year-old Caucasian woman presented with a month history of left temporal headache (with no relieving factors), fatigue, and a weight loss of 4 kilograms. Over the last 5 days patient also experienced progressive visual disturbances: gray and blurred vision in patient’s left eye, with no pain, and an impression of a loss of the inferior half of the left eye vision field.
The right eye was asymptomatic. The patient had no other neurologic signs or symptoms and denied diplopia. She did not have a prior history of headaches. There was no prior history of headaches, smoking, or alcohol consumption. The past medical history was notable for hypertension and osteoporosis. No signs of atherosclerosis were present. An allergy to iodinated contrast was documented. The patient had never undergone any surgery, including spinal procedures.
Examination revealed a tenderness around the left temple, with no prominent vessel visible. Patient’s decimal visual acuity was 0.9 in the right eye and 0.7 in the left eye. The pupillary examination showed relative afferent pupillary defect (RAPD) in the left eye. The optical coherence tomography showed normal retinal nerve fiber layer in both eyes. Ocular motility was full. Fundus examination was normal. There was no ptosis or anisocoria. Intraocular pressure was 18 mmHg in the right eye and 17 mmHg in the left eye. Visual field examination showed inferior altitudinal defect in the left eye. Based on the above-mentioned findings, the diagnosis of PION was made.
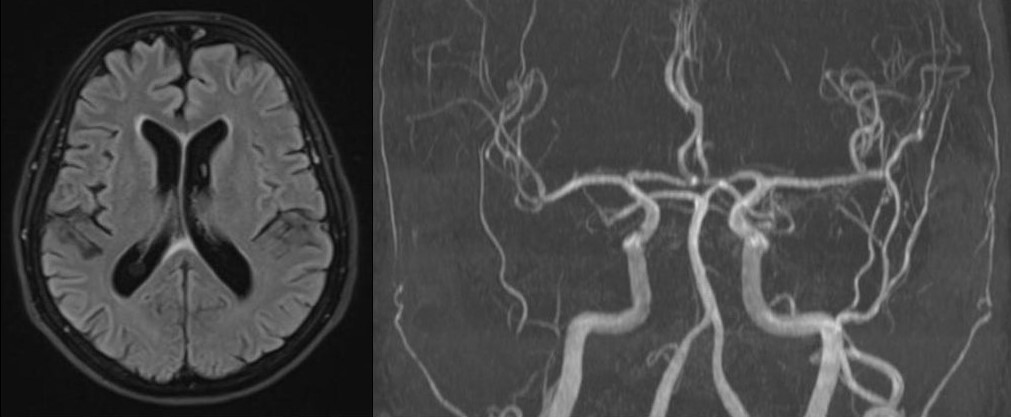
Head magnetic resonance angiography (MRA) and venography with contrast were within normal limits (Fig. 2). Erythrocyte sedimentation rate and CRP levels were normal. The halo sign on both temporal and axillary artery ultrasound was absent. The Southend Giant Cell Arteritis Probability Score (GCAPS) score was 15 [1].
Inflammatory, infectious, and thrombophilic investigations, including syphilis, acquired immune deficiency syndrome, coronavirus disease 2019, varicella zoster virus infection, myelin oligodendrocyte glycoprotein antibody-associated disease, neuromyelitis optica spectrum disorders, antiphospholipid syndrome, and factor V Leiden, were all negative.
Given the high clinical suspicion of GCA, GCAPS score 15 (high-risk patients), and the presence of visual symptoms, the patient was immediately started on 1 g of intravenous methylprednisolone daily for 5 days, followed by an oral prednisone taper (60 mg/day) [2]. The symptoms rapidly improved (although the visual field defect remained unchanged).
At this point, the diagnosis of GCA was made as the patient met the 2022 ACR/EULAR classification criteria for GCA. The patient scored 7 points; a score of 6 points at least is needed for the diagnosis of GCA [3] (Fig. 3).
Fig. 3
The 2022 American College of Rheumatology/EULAR classification criteria for giant cell arteritis.
Source: Ponte C, Grayson PC, Robson JC, et al. 2022 American College of Rheumatology/EULAR Classification Criteria for Giant Cell Arteritis. Arthritis Rheumatol 2022; 74: 1881–1889, DOI: 10.1002/art.42325. A license to reprint the figure was obtained from the publisher of Arthritis & Rheumatology, John Wiley & Sons.

However, given the fact that the patient, who had already suffered from osteoporosis, would be put on chronic glucocorticosteroids (GCs), we wanted to be sure of the absolute necessity of the treatment; thus, a left temporal artery biopsy (TAB) was performed to confirm the diagnosis of GCA. Temporal artery biopsy was performed on the seventh day of the GC treatment. Precisely 2 cm of the frontal branch of the left superficial temporal artery were cut out. Histopathological findings were consistent with GCA. Microscopic examination showed numerous lymphocytes and macrophages forming granulomatous inflammation within the arterial media, nearly completely narrowing the lumen of the vessel, and a disruption of internal elastic lamina (Fig. 4).
Fig. 4
Microscopic image of giant cell arteritis. Biopsy specimen of the frontal branch of the left superficial temporal artery. Hematoxylin and eosin stain; objective: 10 ×.
Source: Photo courtesy of prof. dr hab. n. med. Przemysław Majewski, Department of Clinical Pathomorphology, Poznan University of Medical Sciences.
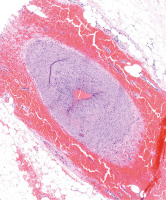
At that point, the patient was classified as having GCA with greater certainty. With the histopathological examination results she scored 12 points (compared to 7 points earlier) when applying the 2022 ACR/EULAR classification criteria for GCA [3].
Discussion
Giant cell arteritis – formerly known as Horton’s disease – is the most common form of systemic vasculitis in patients aged over 50 years. Giant cell arteritis can be defined by multifocal granulomatous arteritis that affects medium- and large-sized blood vessels, with a predisposition to affect the cranial and extracranial arteries [2]. Typical symptoms of GCA include headache (hence such patients are often first seen by neurologists), constitutional symptoms, scalp tenderness, jaw or tongue claudication and various visual disturbances, which are commonly the reason for a referral to ophthalmologists. Giant cell arteritis often co-occurs with polymyalgia rheumatica (PMR). Nearly half of people with GCA have symptoms of PMR, while up to one-fifth of patients with PMR will be diagnosed one day with GCA [4].
Unfortunately, raised ESR and CRP levels, halo sign on temporal artery ultrasound (a hypoechoic, non-compressible, circumferential area around the vessel lumen standing for the vessel wall inflammation) [5] and A-AION are customarily considered by clinicians as indispensable elements to make a diagnosis of GCA, when, in fact, the only absolute requirement to make a diagnosis is age ≥ 50 years at the time of the diagnosis (see the 2022 ACR/EULAR classification criteria for GCA [3], Fig. 3). Our patient had absolutely normal ESR and CRP levels, absent halo sign and PION, not – A-AION. Let us highlight below some key points to clarify why this case was so unusual – and usual – at once.
Not all patients with GCA have elevated ESR and CRP levels. Although it is considered rare, some patients, even with a biopsy-proven GCA, have absolutely normal ESR and CRP levels. Inflammatory markers are helpful in the diagnosis of GCA, but normal values do not exclude the diagnosis. In the very recent retrospective population-based cross-sectional study by Castillejo Becerra et al. [6], normal ESR and CRP levels were observed in 3% of biopsy-proven GCA patients. Combined ESR and CRP levels had a sensitivity of 77% and specificity of 54%. What is more, elevated ESR and CRP levels are not necessary for the diagnosis of GCA according to the 2022 American College of Rheumatology/EULAR Classification Criteria for Giant Cell Arteritis [3].
Although the most recent systematic literature review confirmed very good sensitivity (67%) and specificity (95%) of the halo sign on the temporal artery ultrasound in the diagnosis of GCA [7], not all patients have it, and its absence does not exclude the diagnosis of GCA. Also, large vessel GCA, mainly affecting the aorta and its branches and often occurring alongside cranial GCA, can occur without cranial arteries involvement. What is more, axillary arteries should also be included in the standard ultrasound examination [2, 3, 5, 8].
Regardless of the growing significance of the ultrasound, a TAB remains the gold-standard test in the diagnosis of GCA. As mentioned above, GCA is multifocal (areas of an inflamed artery may alternate with areas of an unaffected artery) [5, 9]. Therefore, a biopsy specimen that is too short might result in omitting the inflamed segments and misleadingly show only the healthy ones (referred to as “skip lesions”). Chu et al. [10] recommend 1.5–2 cm as the optimal biopsy specimen length, with greater lengths unlikely to provide significant additional diagnostic yield to justify risks associated with surgery, such as – most commonly – damage to the temporal branch of the facial nerve resulting in peripheral facial nerve palsy. Moreover, GC treatment can affect the histopathological findings of TAB. Although some references suggest that TAB can remain positive (histopathological findings of the biopsy specimen consistent with GCA) up to 4 or 6 weeks, or even up to 12 months from the GC treatment initiation, TAB should ideally be performed within 2 weeks to avoid a false-negative biopsy [2, 11]. Our patient had precisely 2 cm of the frontal branch of the left superficial temporal artery cut out on the 7th day of the CG treatment. Hereby, we recommend that while suspecting GCA a TAB should be performed ideally within 2 weeks of the initiation of the GC treatment and 2 cm length of the frontal branch of the superficial temporal artery should be biopsied.
Importantly, if the TAB result is negative (which was not the case in our patient), it should be performed on the contralateral side in order to fully rule out the disease. Agostina et al. [12] reported that in 25% of bilateral biopsy cases that were positive only on one side, patients had unilateral temporal arteritis symptoms that were opposite to the side positive for temporal arteritis. Missing diagnosis and premature interruption of GC treatment can lead to disastrous complications, such as visual loss. In order to avoid such situations, some centers perform simultaneous bilateral TAB from the outset.
Biopsy specimen length, right timing of the TAB, uni- or bilaterality of the procedure, and also the number of sections evaluated by the pathologist all matter given the patchy involvement of the temporal arteries in GCA [7].
Posterior ischemic optic neuropathy is an acute optic neuropathy due to ischemia in the posterior portion of the optic nerve (retrobulbar, behind the lamina cribrosa) which is supplied by a pial capillary plexus that is derived from collateral branches of the ophthalmic artery. Posterior ischemic optic neuropathy is a diagnosis of exclusion and is clinically characterized by acute, painless vision loss in one or both eyes, the presence of a RAPD in unilateral or bilateral (but asymmetric) cases, and a normal fundus (including a normal optic disc with no disc edema). In fluorescein angiography a choroidal perfusion deficit is expected. Etiologically, PION can be classified into 3 types: arteritic PION (due to GCA), nonarteritic PION (due to causes other than GCA), and surgical PION (these patients discover dramatic visual loss as soon as they are alert enough after a prolonged, surgical procedure under general anesthesia, e.g. spine surgery or cardiac surgery; surgical PION usually tends to cause bilateral severe visual loss or even complete blindness that is usually permanent) [13]. In PION diffusion-weighted imaging sequences on orbital magnetic resonance imaging (MRI) may show diffusion restriction in the posterior part of the optic nerve with decreased apparent diffusion coefficient indicating ischemic injury [14, 15]. This should encourage clinicians to refer patients for orbital MRI if PION is suspected or when the diagnosis is uncertain.
Although A-AION is the most common ocular manifestation of GCA, other neuro-ophthalmic signs should not dissuade the clinicians from making a diagnosis of GCA. Neuro-ophthalmic manifestations of GCA can be very diverse and also include: PION [4, 16], amaurosis fugax [4, 17], diplopia (attributed to oculomotor, trochlear and abducens nerves palsies or extraocular muscle ischemia) [4, 17–19], paracentral acute middle maculopathy [5, 20, 21], Horner syndrome [22], ischemic mydriasis [4], tonic pupils [4], cotton wool spots [4], non-embolic central retinal artery occlusion, cilioretinal artery occlusion, branch retinal artery occlusion, choroidal artery occlusion, anterior segment ischemia [4], ophthalmic artery occlusion [23, 24], choroidal ischemia [25], ocular ischemic syndrome [4], homonymous hemianopia due to ischemia of the occipital lobe (with the involvement of the vertebral artery) [17] and – very rarely – peripheral ulcerative keratitis, scleritis, and orbital pseudotumour (GCA mimicking nonspecific orbital inflammation) [4].
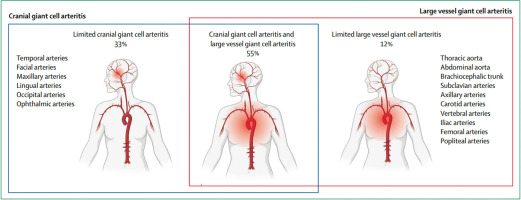
As mentioned earlier, GCA affects not only cranial arteries (temporal, facial, maxillary, lingual, occipital, and ophthalmic arteries) and extracranial arteries (carotid, vertebral arteries), but also larger ones, such as the thoracic aorta, abdominal aorta, brachiocephalic trunk, and subclavian, axillary, iliac, femoral, and popliteal arteries [8]. Thus, GCA can also present as, e.g., aortic aneurysm and dissection, myocardial infarction [26], or stroke [27]. Van der Geest et al. [8] estimated the prevalence of GCA subtypes in a cohort of 241 patients. Of the patients, 33% had limited cranial GCA, 12% had limited large vessel GCA, and 55% had mixed cranial GCA and large vessel GCA (Fig. 5). Therefore, not only ultrasound of temporal and axillary arteries, but also head MRA, [18F]-fluorodeoxyglucose positron emission tomography and computed tomography angiography of the head and body all play an important role in the diagnosis, management, and treatment monitoring of all GCA subtypes [2–4, 7, 8]. Large vessel imaging may provide additional evidence of the disease when the diagnosis is uncertain, especially following a negative TAB result [2]. For more detailed information, see EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice: 2023 update [28].
When GCA is strongly suspected, GC treatment should be started immediately without waiting for the full diagnostic process to be completed [29].
To the best of our knowledge, there have been only 39 reports in the literature of PION secondary to GCA, all collected in the papers by Albarrak et al. [30], Nichani et al. [31], Arai et al. [32], and Yan et al. [33], but none of them have identical features to ours.
Conclusions
Giant cell arteritis can present very diversely, and although it is believed to be a purely rheumatological entity that affects only temporal arteries, in reality it affects multiple, often larger, arteries and demands an interdisciplinary approach. Unquestionably, GCA is a disease that requires immediate medical recognition to initiate treatment promptly, since overlooking the diagnosis may have catastrophic consequences. With such clear diagnostic criteria, accessible specialist diagnostic tools, and informative management guidelines, diagnostic certainty is nowadays easy to reach [4].
Fig. 5
Distribution of large vessels and cranial arteries involvement in giant cell arteritis.
Source: van der Geest KSM, Sandovici M, Bley TA, et al. Large vessel giant cell arteritis. Lancet Rheumatol 2024; 6: e397–e408, DOI: 10.1016/S2665-9913(23)00300-4. A license to reprint the figure was obtained from Kornelis S. M. van der Geest and the publisher of The Lancet Rheumatology, Elsevier.