Introduction
Giant cell arteritis (GCA) is a granulomatous large vessels (LV) vasculitis that affects both cranial and extracranial arteries. It is considered the most common non-infectious vasculitis in older people [1]. Giant cell arteritis can be associated with polymyalgia rheumatica (PMR), which in turn is considered the most common inflammatory rheumatic disease in populations aged over 70 years. Patients with GCA have manifestations of PMR about in 40–60% of cases, whereas patients with PMR have manifestations of GCA in < 20% of cases.
Recently, some researchers proposed the term GCA-PMR spectrum disease (GPSD), considering these two diseases as a continuum [2]. Without a doubt, isolated PMR exists. The possibility that GCA and PMR are concurrent diseases rather than a common spectrum of inflammatory diseases has recently been re-discussed [3]. The association of GCA with PMR has significant therapeutic and prognostic consequences [4, 5].
In some patients with PMR, GCA is subclinical, as underlying inflammation of LV is present without evidence of related clinical manifestations. A recent systematic review and meta-analysis of individual patient data concluded that circa 25% of patients with PMR have subclinical GCA when screened by positron emission tomography/total body computed tomography with fluorine 18-fluorodeoxyglucose (18F-FDG PET/CT) [6].
More recently, a cross-sectional international study reported that > 20% of consecutive, non-selected PMR patients had ultrasound (US) findings consistent with subclinical GCA [7].
Therefore, PMR could be a signal for an underlying subclinical GCA. The presence of subclinical GCA has been documented to be associated with a greater risk of PMR relapse, slower minor response to glucocorticosteroids (GCs) and prolonged treatment time, more frequent atypical manifestations, and higher rates of ischemic complications during follow-up [8–10]. Accordingly, recognition of subclinical GCA is very important in everyday clinical practice.
Different factors have been proposed as predictive of subclinical GCA in PMR patients. To date, the published literature reports scant data about the risk of subclinical GCA when compared to duration of morning stiffness (MS) in patients with newly diagnosed PMR. Long-lasting MS is listed in all diagnostic or classification criteria proposed for PMR over the years.
According to the classification criteria proposed in 2012 by the European League Against Rheumatism (now European Alliance of Associations for Rheumatology)/American College of Rheumatology (EULAR/ACR) collaborative study, MS duration is > 45 min [11]. Recently, another EULAR/ACR collaborative study proposed the addition of long-lasting MS in shoulders/neck to the existing classification criteria for GCA [12].
Given this background, the aim of this study was to assess the association between subclinical GCA and long-lasting MS in patients with newly diagnosed PMR.
Material and methods
The authors performed an observational, retrospective, single-centre cohort study of patients referred to our public out-of-hospital rheumatologic clinic between January 2015 and December 2020, who could be classified as having PMR according to the 2012 EULAR/ACR criteria. Specifically, all the enrolled patients had a total score of 5 or more using the EULAR/ACR clinical and US criteria (Table I).
Table I
EULAR/ACR classification criteria for polymyalgia rheumatica [11, mod.]
In accordance with the provisions of the Italian National Health System, all patients were referred to our clinic by their general practitioner. The study was performed in Sant’Agnello (Health District no. 59, Azienda Sanitaria Locale Napoli 3, Italy). The basic inclusion and exclusion criteria have been previously reported [13].
In short, we assessed the following variables: sex, age at onset, presence or absence of MS duration longer than 45 min, routine analyses, chest X-ray, abdominal US examination, body mass index (BMI), patient’s numerical Visual Analogue Scale (VAS) score.
In particular, the patient’s VAS was from 0 to 100. Routine analyses also included rheumatoid factor (RF), anti-citrullinated protein antibodies (ACPA), anti-nuclear antibodies (ANA), and anti-neutrophil cytoplasmic antibodies (ANCA).
Regarding the definition of MS, we used that proposed by Lineker et al. [14] for patients with rheumatoid arthritis (RA): “slowness or difficulty moving the joints when getting out of bed or after staying in one position too long, which involves both sides of the body and gets better with movement”. As highlighted by a report from the OMERACT (informally organized network aimed at improving outcome measurement in rheumatology) 2016 Stiffness Special Interest Group, qualitative investigation of MS in patients with RA is consistent with qualitative research studies on MS performed in patients with PMR [15]. We assessed MS in the shoulder and neck in the past 24 hours, in a dichotomous way (Yes if> 45 min/No if < 45 min).
According to the aim of this study, we specifically assessed the association between MS > 45 min and presence of subclinical GCA. Subclinical GCA was investigated through US examination of a core set of arteries (temporal, axillary, common carotid, and subclavian arteries). All the US examinations were performed by two specialists with 10 years’ expertise in vascular US, in accordance with the EULAR recommendations for the use of imaging in LV vasculitis [16].
Consequently, we used a high frequency linear US probe > 18 MHz for temporal arteries and a probe ranging from 6 to 15 MHz for axillary, carotid and subclavian arteries. Doppler pulse repetition frequency was 2–3.5 kHz for temporal arteries and 3–4 kHz for the other arteries. The colour box had an angle correction between angle waves and arteries of < 60°. Finally, the B-mode gain was adjusted to fill only the lumen of the explored arteries, avoiding anechoic appearance of the artery wall.
Patients who did not have GCA symptoms but showed a halo sign in at least one of these arteries were assessed as having subclinical GCA and enrolled in this study. Doubtful cases were excluded after discussion.
We did not include PMR patients who had the following clinical manifestations: new-onset headache, jaw claudication, sudden visual disturbances, temporal artery tenderness, or widespread scalp tenderness, according to the 1990 ACR criteria for temporal arteritis [17]. The association between MS duration and presence of clinical GCA, in fact, was not the focus of our study.
Statistical analysis
The normality of continuous variables was assessed using the Kolmogorov-Smirnov test. We reported all the descriptive data of normally distributed variables as the mean ±standard deviation (SD) or, in the case of non-normal distribution, as the median and interquartile range; binary data were reported as percentages. Differences between groups were compared using an unpaired Student’s t-test when clinical and biological data were expressed as continuous variables, and the χ2 test for categorical variables.
The presence of underlying subclinical GCA, the outcome of our interest, was assessed using a logistic regression analysis, adjusted for potential covariates. The factors included were tested for a significant difference between MS+ and MS– or associated with the outcome of interest in the univariate analyses, taking a p-value < 0.10.
The collinearity among covariates was assessed using the variance inflation factor (VIF) using a threshold of 2 as a reason for exclusion, but no factor was excluded for this reason. The results were then reported as odds ratios (ORs) with their 95% confidence intervals (CIs).
All the datasets were analysed using a standard statistical package (SPSS Statistics for Windows version 21.0, IBM, USA) and a p-value < 0.05 was considered as statistically significant.
Results
We included in our study 143 patients (35 men and 108 women). Their median age was 71.5 years. Thirty-five had MS duration < 45 min at the time of PMR diagnosis. Table II lists their main demographic, clinical and laboratory data by presence (MS-Yes) or not (MS-No) of MS < 45 min.
Table II
Baseline characteristics, by presence or not of morning stiffness
Participants with or without MS < 45 min did not differ in terms of mean age (p = 0.67) or female sex (p = 0.44). Patients with MS < 45 min had significantly higher serum inflammatory parameter levels and they did not differ in terms of the other characteristics examined.
Prevalence of subclinical GCA was 16.1%. Briefly, subclinical GCA was found in 23 PMR patients: 18 had a cranial and 5 an extracranial GCA. Presence of MS < 45 min at the time of PMR diagnosis was significantly associated, both in univariate and in multivariable analysis, with a very low presence of subclinical GCA.
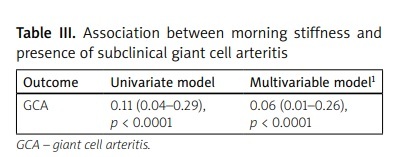
In particular, in univariate analysis, we found that MS < 45 min was associated with a lower prevalence of GCA (OR = 0.11, 95% CI: 0.04–0.29; p < 0.0001). A multivariable analysis with all the other assessed covariates confirmed a very low association (OR = 0.06, 95% CI: 0.01–0.26; p < 0.0001) (Table III).
Table III
Association between morning stiffness and presence of subclinical giant cell arteritis
| Outcome | Univariate model | Multivariable model1 |
|---|---|---|
| GCA | 0.11 (0.04–0.29), p < 0.0001 | 0.06 (0.01–0.26), p < 0.0001 |
1 Multivariable model included, as covariates: age, sex, serum levels of C reactive protein and fibrinogen, body mass index, Visual Analogue Scale. The data are reported as odds ratios (ORs) with their correspondent 95% confidence intervals (CIs) and p-values for the association between morning stiffness and the different forms (cranial and extracranial) of GCA.
Discussion
In our retrospective study, an MS < 45 min at the time of PMR diagnosis in patients without GCA clinical manifestations was associated with a significantly lower risk of subclinical GCA, when patients were screened by US, of approximately 90%.
Different factors have been proposed as predictive of subclinical GCA in PMR patients. For instance, whilst higher erythrocyte sedimentation rate (ESR) and/or C-reactive protein (CRP) concentrations in PMR patients with underlying subclinical GCA have been observed [18], other diagnostic criteria should be considered in everyday clinical practice, since ESR and CRP do not allow differential diagnosis [19].
The association between duration of MS and subclinical GCA in patients with newly diagnosed PMR has been poorly assessed in the published literature. The reported results are conflicting. For instance, a prospective, multicentre, international cohort study of 346 consecutive non-selected PMR patients found a longer duration of MS in the PMR patients with subclinical GCA compared with the patients without PMR and subclinical GCA (mean 72.3 vs. 34.5 min; p < 0.001) [7].
In contrast, a systematic review’s univariable analysis of 13 studies reporting on 566 GC-naive patients with PMR did not observe a significant association between duration of MS and subclinical GCA [6]. Other studies did not assess this potential association [20, 21].
The possibility that PMR patients with MS < 45 min could be a subset of disease characterised by very low risk of underlying GCA is an intriguing working hypothesis that deserves further investigation. We classified 35 patients as having PMR even if MS duration was < 45 min.
According to the 2012 EULAR/ACR study, an MS duration > 45 min had an odds ratio (OR) of 5 when a combination of clinical and US criteria was used, and of 6.2 when only clinical criteria were used. These OR values were much higher than those attributed to all other proposed classification criteria.
Nevertheless, according to these same criteria, PMR categorization is possible even if MS > 45 min is absent, provided that the other proposed criteria are satisfactorily met [11]. Consequently, we were authorised to describe 35 patients as having PMR because their total score was 5 or more using the EULAR/ACR clinical and US criteria.
Compared to the aforementioned studies, the prevalence of subclinical GCA in newly diagnosed PMR patients was relatively low in our cohort (16.1% vs. > 20%). This could be due to PMR being commonly managed in out-of-hospital practice with a referral to hospital when serious diagnostic difficulties are present [22–25].
Therefore, a referral bias introduced by selection of less critical patients cannot be excluded in our study. On the other hand, in our experience, routine US screening for subclinical GCA is considered standard of care in all patients where PMR diagnosis has been confirmed because of prognostic repercussions and therapeutic planning. To date, we have failed to give a convincing explanation of why the risk of subclinical GCA was very low when MS duration was reported as < 45 min.
Interestingly, mean ESR values and CRP concentrations were significantly higher in our group of PMR patients with MS > 45 min when compared to the group of PMR patients with MS < 45 min. In this latter group, we found a significantly low risk of subclinical GCA, when patients were screened by US, of approximately 90%. This would indirectly confirm the limited value of higher ESR and CRP as risk factors for underlying subclinical GCA.
The putative role of other, different laboratory markers such as angiopoietin-2 in indicating an underlying GCA in this group of PMR patients could be considered [26]. However, the assessment of angiopoietin-2 serum levels is not a routine test in everyday clinical practice, as US examinations are preferred due to their fast response time.
Study limitations
We acknowledge that our research has limitations, including all known limits of a retrospective out-of-hospital study. In particular, our study might suffer from inclusion bias: we cannot rule out that some patients did not suffer from isolated aortitis at the time of PMR diagnosis [27–31].
Indeed, it is widely considered that magnetic resonance imaging (MRI) and/or PET/CT are particularly useful at sites where US is difficult to perform due to the restricted acoustic window [32, 33]. Only a few patients had MRI and/or PET/CT scans (more accurate diagnostic tools): in some cases, patients denied consent; in other cases, there were technical or bureaucratic difficulties (our health district has limited in-house imaging services, and MRI and PET/CT scans are outsourced).
In addition, the very low number of PMR patients with subclinical extracranial GCA (only five) did not allow the enrolled patients to be stratified according to the type of cranial or extracranial involvement. Finally, we evaluated MS duration dichotomously (Yes/No), and therefore we could not assess the correlation between risk of subclinical GCA and MS duration in minutes. Finally, we evaluated MS duration dichotomously (Yes/No), and therefore we could not assess the correlation between risk of subclinical GCA and MS duration in minutes. On the other hand, our study has strengths. Firstly, there was rigorous selection of patients.
Additionally, we assessed PMR patients referred to a public out-of-hospital rheumatologic clinic, and this can be another strength. All previous studies were indeed based on data from hospital and/or university databases. Yet, PMR is commonly managed in out-of-hospital practice [34].
Therefore, inclusion bias cannot be ruled out in databases from the so-called second- or third-level centres, where only PMR patients with diagnostic and/or therapeutic critically are usually referred. To the best of our knowledge, this is the first study that assessed data from PMR patients referred to a public, out-of-hospital rheumatologic clinic. Greater input by the public out-of-hospital rheumatologists is needed in studies on PMR and GCA, but their active involvement is still lacking in the published literature [35–38].
Conclusions
There is very little literature on the association between presence of subclinical GCA and the MS duration reported by patients with newly diagnosed PMR. Data about this association are conflicting.
Our study highlighted that an MS < 45 min at the time of isolated PMR diagnosis was associated with a significantly low risk of subclinical GCA, when patients were screened by US, of approximately 90%.
A subsequent prospective study could focus on the identification of an MS cut-off value that can improve sensitivity and specificity for subclinical GCA in patients with newly diagnosed PMR.