Current issue
Archive
Online first
About the Journal
Editorial Office
Editorial Board
Publisher
Editorial Policies
Ethical standards and procedures
Abstracting and indexing
Reviewers
Honorary Reviewers
Subscription
Contact
Most read articles
Instructions for authors
Article processing charge (APC)
Books and Events
Books
Events
ORIGINAL PAPER
Distinct profiles of circulating lymphocytes reflect clinical and serological variations in idiopathic inflammatory myopathies
1
Department of Rheumatology, Medical University of Lodz, Poland
Submission date: 2025-02-08
Final revision date: 2025-05-10
Acceptance date: 2025-10-22
Publication date: 2026-04-30
Reumatologia 2026;64(2):94-104
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Idiopathic inflammatory myopathies (IIM) are a heterogeneous group of autoimmune diseases characterized by muscle inflammation and systemic involvement. The study aimed to evaluate the immunophenotypic profile of circulating lymphocytes in patients with IIM and investigate its association with clinical manifestations, serological profiles, treatment regimens, and disease outcomes.
Material and methods:
This single-center, cross-sectional study included 40 patients with IIM and 5 age- and sex-matched healthy controls. Peripheral blood samples were collected and processed to isolate peripheral blood mononuclear cells using the density gradient centrifugation method. Flow cytometry analysis was performed to phenotype lymphocyte subpopulations, including T cells (CD3+CD4+ and CD3+CD8+), B cells (CD3–CD19+), and natural killer (NK) cells (CD3–CD16+CD56+). Statistical analysis was conducted to compare lymphocyte profiles between the study and control groups and to assess correlations with clinical features, serological markers, and treatment regimens.
Results:
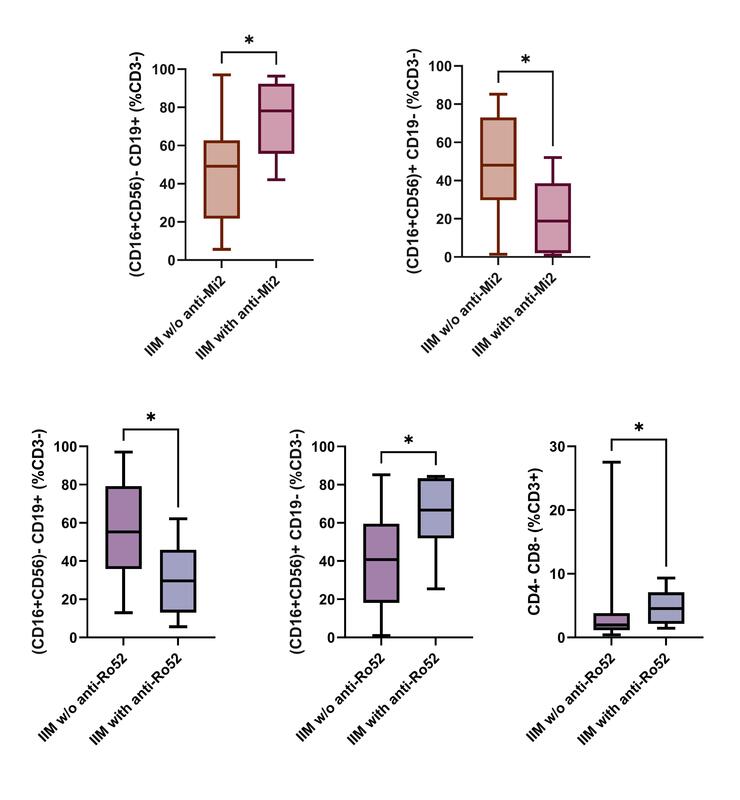
The IIM group had a mean age of 58.55 ±12.70 years, with a predominance of females (60%). The most common IIM subtypes were antisynthetase syndrome (35%) and dermatomyositis (30%). No significant differences in the profiles of circulating lymphocytes were found between IIM patients and healthy controls. Patients with cardiac involvement had significantly higher proportions of double-positive T cells (p = 0.0035), while those with cutaneous ulcers and dysphagia showed significantly lower proportions of CD4+CD8+ T cells. Natural killer cells were significantly lower in patients with anti-Mi2 antibodies (p = 0.0155) but higher in anti-Ro52 positive patients (p = 0.0150). In contrast, B cells were notably higher in patients with anti-Mi2 (p = 0.0182) and lower in individuals with anti-Ro52 (p = 0.0124). Patients with anti-Ro52 positivity were also characterized by higher proportions of double-negative T cells (p = 0.0465). Treatment regimens did not impact the profiles of circulating lymphocytes.
Conclusions:
Patients with IIM exhibit distinct alterations in lymphocyte subpopulations, with specific immune cell profiles associated with clinical phenotypes and serological profiles.
Idiopathic inflammatory myopathies (IIM) are a heterogeneous group of autoimmune diseases characterized by muscle inflammation and systemic involvement. The study aimed to evaluate the immunophenotypic profile of circulating lymphocytes in patients with IIM and investigate its association with clinical manifestations, serological profiles, treatment regimens, and disease outcomes.
Material and methods:
This single-center, cross-sectional study included 40 patients with IIM and 5 age- and sex-matched healthy controls. Peripheral blood samples were collected and processed to isolate peripheral blood mononuclear cells using the density gradient centrifugation method. Flow cytometry analysis was performed to phenotype lymphocyte subpopulations, including T cells (CD3+CD4+ and CD3+CD8+), B cells (CD3–CD19+), and natural killer (NK) cells (CD3–CD16+CD56+). Statistical analysis was conducted to compare lymphocyte profiles between the study and control groups and to assess correlations with clinical features, serological markers, and treatment regimens.
Results:
The IIM group had a mean age of 58.55 ±12.70 years, with a predominance of females (60%). The most common IIM subtypes were antisynthetase syndrome (35%) and dermatomyositis (30%). No significant differences in the profiles of circulating lymphocytes were found between IIM patients and healthy controls. Patients with cardiac involvement had significantly higher proportions of double-positive T cells (p = 0.0035), while those with cutaneous ulcers and dysphagia showed significantly lower proportions of CD4+CD8+ T cells. Natural killer cells were significantly lower in patients with anti-Mi2 antibodies (p = 0.0155) but higher in anti-Ro52 positive patients (p = 0.0150). In contrast, B cells were notably higher in patients with anti-Mi2 (p = 0.0182) and lower in individuals with anti-Ro52 (p = 0.0124). Patients with anti-Ro52 positivity were also characterized by higher proportions of double-negative T cells (p = 0.0465). Treatment regimens did not impact the profiles of circulating lymphocytes.
Conclusions:
Patients with IIM exhibit distinct alterations in lymphocyte subpopulations, with specific immune cell profiles associated with clinical phenotypes and serological profiles.
REFERENCES (28)
1.
Lundberg IE, Fujimoto M, Vencovsky J, et al. Idiopathic inflammatory myopathies. Nat Rev Dis Prim 2021; 7: 86, DOI: 10.1038/ s41572-021-00321-x.
2.
Oldroyd A, Lilleker J, Chinoy H. Idiopathic inflammatory myopathies – a guide to subtypes, diagnostic approach and treatment. Clin Med J R Coll Physicians London 2017; 17: 322–328, DOI: 10.7861/clinmedicine.17-4-322.
3.
Grundtman C, Malmström V, Lundberg IE. Immune mechanisms in the pathogenesis of idiopathic inflammatory myopathies. Arthritis Res Ther 2007; 9: 208, DOI: 10.1186/ar2139.
4.
Franco C, Gatto M, Iaccarino L, et al. Lymphocyte immunophenotyping in inflammatory myositis: A review. Curr Opin Rheumatol 2021; 33: 522–528, DOI: 10.1097/BOR.0000000000000831.
5.
Miller FW, Love LA, Barbieri SA, et al. Lymphocyte activation markers in idiopathic myositis: Changes with disease activity and differences among clinical and autoantibody subgroups. Clin Exp Immunol 1990; 81: 373–379, DOI: 10.1111/j.1365-2249.1990.tb05341.x.
6.
O’Gorman MRG, Corrochano V, Roleck J, et al. Flow cytometric analyses of the lymphocyte subsets in peripheral blood of children with untreated active Juvenile dermatomyositis. Clin Diagn Lab Immunol 1995; 2: 205–208, DOI: 10.1128/cdli. 2.2.205-208.1995.
7.
Sasaki H, Takamura A, Kawahata K, et al. Peripheral blood lymphocyte subset repertoires are biased and reflect clinical features in patients with dermatomyositis. Scand J Rheumatol 2019; 48: 225–229, DOI: 10.1080/03009742.2018.1530371.
8.
Espinosa-Ortega F, Gómez-Martin D, Santana-De Anda K, et al. Quantitative T cell subsets profile in peripheral blood from patients with idiopathic inflammatory myopathies: Tilting the balance towards proinflammatory and pro-apoptotic subsets. Clin Exp Immunol 2015; 179: 520–528, DOI: 10.1111/cei.12475.
9.
Hou X, Yang C, Lin M, et al. Altered peripheral helper T cells in peripheral blood and muscle tissue of the patients with dermatomyositis. Clin Exp Med 2021; 21: 655–661, DOI: 10.1007/s10238-021-00713-z.
10.
Antiga E, Kretz CC, Klembt R, et al. Characterization of regulatory T cells in patients with dermatomyositis. J Autoimmun 2010; 35: 342–350, DOI: 10.1016/j.jaut.2010.07.006.
11.
Wang H, Lan L, Wang J, et al. Alterations of B-Cell subsets in Peripheral Blood from Adult Patients with Idiopathic Membranous Nephropathy. Immunol Lett 2024; 266, DOI: 10.1016/j.imlet. 2024.106838.
12.
Reyes-Huerta RF, Mandujano-López V, Velásquez-Ortiz MG, et al. Novel B-cell subsets as potential biomarkers in idiopathic inflammatory myopathies: Insights into disease pathogenesis and disease activity. J Leukoc Biol 2024; 116: 84–94, DOI: 10.1093/jleuko/qiae083.
13.
Li W, Tian X, Lu X, et al. Significant decrease in peripheral regulatory B cells is an immunopathogenic feature of dermatomyositis. Sci Rep 2016; 6: 27479, DOI: 10.1038/srep27479.
14.
Viguier M, Fouéré S, La Salmonière P De, et al. Peripheral blood lymphocyte subset counts in patients with dermatomyositis clinical correlations and changes following therapy. Medicine (Baltimore) 2003; 82: 82–86, DOI: 10.1097/00005792-200303000-00002.
15.
Nelke C, Pawlitzki M, Schroeter CB, et al. High-Dimensional Cytometry Dissects Immunological Fingerprints of Idiopathic Inflammatory Myopathies. Cells 2022; 11, DOI: 10.3390/cells11203330.
16.
Dzangué-Tchoupou G, Allenbach Y, Preuße C, et al. Mass cytometry reveals an impairment of B cell homeostasis in anti- synthetase syndrome. J Neuroimmunol 2019; 332: 212–215, DOI: 10.1016/j.jneuroim.2019.04.014.
17.
Ishii W, Matsuda M, Shimojima Y, et al. Flow cytometric analysis of lymphocyte subpopulations and Th1/Th2 balance in patients with polymyositis and dermatomyositis. Intern Med 2008; 47: 1593–1599, DOI: 10.2169/internalmedicine.47.0967.
18.
Wang Y, Li Q, Lv X, et al. Peripheral Th17/Treg imbalance in Chinese patients with untreated antisynthetase syndrome associated interstitial lung disease. Int Immunopharmacol 2024; 138: 112403, DOI: 10.1016/j.intimp.2024.112403.
19.
Wang K, Zhao J, Chen Z, et al. CD4+ CXCR4+ T cells as a novel prognostic biomarker in patients with idiopathic inflammatory myopathy-associated interstitial lung disease. Rheumatol (United Kingdom) 2019; 58: 511–521, DOI: 10.1093/rheumatology/ key341.
20.
Wang DX, Lu X, Zu N, et al. Clinical significance of peripheral blood lymphocyte subsets in patients with polymyositis and dermatomyositis. Clin Rheumatol 2012; 31: 1691–1697, DOI: 10.1007/s10067-012-2075-4.
21.
Pawlitzki M, Nelke C, Rolfes L, et al. Nk cell patterns in idiopathic inflammatory myopathies with pulmonary affection. Cells 2021; 10: 2551, DOI: 10.3390/cells10102551.
22.
Parel Y, Chizzolini C. CD4+ CD8+ double positive (DP) T cells in health and disease. Autoimmun Rev 2004; 3: 215–220, DOI: 10.1016/j.autrev.2003.09.001.
23.
Lin S, Zhang Y, Cao Z, et al. Decrease in cell counts and alteration of phenotype characterize peripheral NK cells of patients with anti-MDA5-positive dermatomyositis. Clin Chim Acta 2023; 543: 117321, DOI: 10.1016/j.cca.2023.117321.
24.
Guo Y, Liu H, Chen B, et al. Dysregulated CD38 expression on T cells was associated with rapidly progressive interstitial lung disease in anti-melanoma differentiation-associated gene 5 positive dermatomyositis. Front Immunol 2024; 15: 1455944, DOI: 10.3389/fimmu.2024.1455944.
25.
Betteridge Z, Tansley S, Shaddick G, et al. Frequency, mutual exclusivity and clinical associations of myositis autoantibodies in a combined European cohort of idiopathic inflammatory myopathy patients. J Autoimmun 2019; 101: 48–55, DOI: 10.1016/j.jaut.2019.04.001.
26.
Wu Z, Zheng Y, Sheng J, et al. CD3+CD4-CD8- (Double-Negative) T Cells in Inflammation, Immune Disorders and Cancer. Front Immunol 2022; 13: 816005, DOI: 10.3389/fimmu.2022.816005.
27.
Li H, Tsokos GC. Double-negative T cells in autoimmune diseases. Curr Opin Rheumatol 2021; 33: 163–172, DOI: 10.1097/BOR.0000000000000778.
28.
Ernste FC, Crowson CS, De Padilla CL, et al. Longitudinal peripheral blood lymphocyte subsets correlate with decreased disease activity in juvenile dermatomyositis. J Rheumatol 2013; 40: 1200–1211, DOI: 10.3899/jrheum.121031.
Copyright: © Narodowy Instytut Geriatrii, Reumatologii i Rehabilitacji w Warszawie. This is an Open Access journal, all articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0) License (https://creativecommons.org/licenses/by-nc-sa/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, provided the original work is properly cited and states its license.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.