Bieżący numer
Archiwum
Online first
O czasopiśmie
Redakcja
Rada Naukowa
Wydawca
Standardy etyczne i procedury
Bazy indeksacyjne
Recenzenci
Recenzenci Honorowi
Prenumerata
Kontakt
Najczęściej czytane artykuły
Dla autorów
Opłata za przetwarzanie artykułu (APC)
Książki i Konferencje
Książki
Konferencje
SARS CoV-2/COVID-19
PRACA ORYGINALNA
A multicentre study for clinical phenotype prediction in juvenile dermatomyositis: categorical principal component analysis-based hierarchical clustering
1
Department of Pediatric Rheumatology, Faculty of Medicine, Dokuz Eylul University, Izmir, Turkey
2
Department of Pediatric Rheumatology, Faculty of Medicine, Hacettepe University, Ankara, Turkey
3
Department of Pediatric Rheumatology, Ankara Etlik Integrated Health Campus, Ankara, Turkey
4
Department of Pediatric Rheumatology, Faculty of Medicine, Istanbul University, Istanbul, Turkey
5
Department of Pediatric Rheumatology, Istanbul University Cerrahpasa Medical School, Istanbul, Turkey
6
Department of Pediatric Rheumatology, Umraniye Research and Training Hospital, University of Health Sciences, Istanbul, Turkey
7
Department of Pediatric Rheumatology, Faculty of Medicine, Uludag University, Bursa, Turkey
8
Department of Pediatric Rheumatology, Faculty of Medicine, Gazi University, Ankara, Turkey
9
Department of Pediatric Rheumatology, Faculty of Medicine, Kocaeli University, Kocaeli, Turkey
10
Department of Pediatric Nephrology and Rheumatology, Faculty of Medicine, Karadeniz Technical University, Trabzon, Turkey
11
Department of Pediatric Rheumatology, Malatya Training and Research Hospital, Malatya, Turkey
12
Department of Pediatric Rheumatology, Istanbul Medeniyet University, Goztepe Prof. Dr. Suleyman Yalcın City Hospital, Istanbul, Turkey
13
Department of Pediatric Rheumatology, Faculty of Medicine, Pamukkale University, Izmir, Turkey
14
Department of Pediatric Rheumatology, University of Health Sciences Dr. Behcet Uz Child Disease and Pediatric Surgery Training and Research Hospital, Izmir, Turkey
15
Department of Biostatistics and Medical Informatics, Dokuz Eylul University Faculty of Medicine, Izmir, Turkey
Data nadesłania: 30-12-2024
Data ostatniej rewizji: 28-05-2025
Data akceptacji: 26-08-2025
Data publikacji: 23-04-2026
Reumatologia 2026;64(2):83-93
SŁOWA KLUCZOWE
DZIEDZINY
STRESZCZENIE
Introduction:
Juvenile dermatomyositis (JDM) is the most common inflammatory myopathy in childhood and exhibits a heterogeneous disease course. This study aimed to analyse and identify phenotypic clusters by examining the laboratory findings, nailfold capillaroscopy results, and myositis- specific autoantibodies (MSAs) in patients with JDM.
Material and methods:
This retrospective cohort study included data from patients with JDM treated at the Paediatric Rheumatology Departments of 14 advanced health centres in Turkey. A categorical principal component analysis (CATPCA)-based hierarchical cluster analysis method was employed for clustering.
Results:
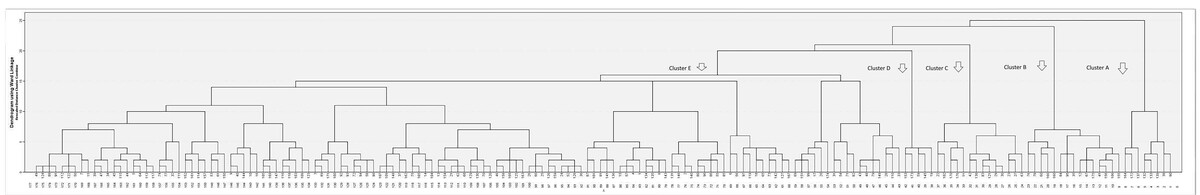
A total of 176 JDM patients were enrolled, and 5 phenotypic clusters were identified using 23 categorical variables. These clusters were interpreted as follows: Cluster A with severe muscle weakness and oesophageal involvement requiring intensive immunosuppressive treatment; Cluster B with amyopathic/hypomyopathic patients; Cluster C with skin manifestations and lung involvement; Cluster D with complicated skin manifestations; and Cluster E with classic JDM. The clinical and laboratory findings and treatments of these 5 clusters were compared. Fatigue, myalgia, photosensitivity, Raynaud’s phenomenon, and the use of pulse glucocorticosteroids, intravenous immunoglobulin, and cyclophosphamide treatments differed between the groups (p < 0.001, p = 0.002, p = 0.015, p = 0.036, p = 0.002, p = 0.006, and p = 0.024, respectively). Myositis-specific autoantibodies results were available for 119 patients (65.3%). The most frequent MSAs were antinuclear matrix protein 2 (26.1%) and anti-transcription intermediary factor 1 (20.9%). However, no significant differences were found in MSAs or nailfold capillaroscopy findings.
Conclusions:
We identified 5 clusters based on patient symptoms and findings. The identification of these 5 clusters can guide more effective treatment strategies in clinical practice. Additionally, these approaches may contribute to improving patients’ quality of life and long-term outcomes by increasing the feasibility of individualised treatment.
Juvenile dermatomyositis (JDM) is the most common inflammatory myopathy in childhood and exhibits a heterogeneous disease course. This study aimed to analyse and identify phenotypic clusters by examining the laboratory findings, nailfold capillaroscopy results, and myositis- specific autoantibodies (MSAs) in patients with JDM.
Material and methods:
This retrospective cohort study included data from patients with JDM treated at the Paediatric Rheumatology Departments of 14 advanced health centres in Turkey. A categorical principal component analysis (CATPCA)-based hierarchical cluster analysis method was employed for clustering.
Results:
A total of 176 JDM patients were enrolled, and 5 phenotypic clusters were identified using 23 categorical variables. These clusters were interpreted as follows: Cluster A with severe muscle weakness and oesophageal involvement requiring intensive immunosuppressive treatment; Cluster B with amyopathic/hypomyopathic patients; Cluster C with skin manifestations and lung involvement; Cluster D with complicated skin manifestations; and Cluster E with classic JDM. The clinical and laboratory findings and treatments of these 5 clusters were compared. Fatigue, myalgia, photosensitivity, Raynaud’s phenomenon, and the use of pulse glucocorticosteroids, intravenous immunoglobulin, and cyclophosphamide treatments differed between the groups (p < 0.001, p = 0.002, p = 0.015, p = 0.036, p = 0.002, p = 0.006, and p = 0.024, respectively). Myositis-specific autoantibodies results were available for 119 patients (65.3%). The most frequent MSAs were antinuclear matrix protein 2 (26.1%) and anti-transcription intermediary factor 1 (20.9%). However, no significant differences were found in MSAs or nailfold capillaroscopy findings.
Conclusions:
We identified 5 clusters based on patient symptoms and findings. The identification of these 5 clusters can guide more effective treatment strategies in clinical practice. Additionally, these approaches may contribute to improving patients’ quality of life and long-term outcomes by increasing the feasibility of individualised treatment.
REFERENCJE (47)
1.
Feldman BM, Rider LG, Reed AM, Pachman LM. Juvenile dermatomyositis and other idiopathic inflammatory myopathies of childhood. Lancet 2008; 371: 2201–2212, DOI: 10.1016/S0140- 6736(08)60955-1.
2.
Sanner H, Gran JT, Sjaastad I, Flatø B. Cumulative organ damage and prognostic factors in juvenile dermatomyositis: a cross-sectional study median 16.8 years after symptom onset. Rheumatology (Oxford) 2009; 48: 1541–1547, DOI: 10.1093/rheumatology/kep302.
3.
Pilkington CA, Feldman BM, Sontichai W. Textbook of Pediatric Rheumatology. 8th ed. Elsevier Inc, Philadelphia 2021; 360–376.
4.
Sordet C, Goetz J, Sibilia J. Contribution of autoantibodies to the diagnosis and nosology of inflammatory muscle disease. Joint Bone Spine 2006; 73: 646–654, DOI: 10.1016/ j.jbspin.2006.04.005.
5.
Bohan A, Peter JB. Polymyositis and dermatomyositis (first of two parts). N Engl J Med 1975; 292: 344–347, DOI: 10.1056/NEJM 197502132920706.
6.
Bohan A, Peter JB. Polymyositis and dermatomyositis (second of two parts). N Engl J Med 1975; 292: 403–407, DOI: 10.1056/NEJM197502202920807.
7.
Smith V, Herrick AL, Ingegnoli F, et al. Standardisation of nailfold capillaroscopy for the assessment of patients with Raynaud’s phenomenon and systemic sclerosis. Autoimmun Rev 2020; 19: 102458, DOI: 10.1016/j.autrev.2020.102458.
8.
Izenman AJ. Modern Multivariate Statistical Techniques: Regression, Classification, and Manifold Learning. Springer, New York, NY 2006; 996.
9.
Kim MJ, Ahn EY, Hwang W, et al. Association between fever pattern and clinical manifestations of adult-onset Still’s disease: unbiased analysis using hierarchical clustering. Clin Exp Rheumatol 2018; 36 (6 Suppl 115): 74–79.
10.
Hässler S, Lorenzon R, Binvignat M, et al. Clinical correlates of lifetime and current comorbidity patterns in autoimmune and inflammatory diseases. J Autoimmun 2024; 149: 103318, DOI: 10.1016/j.jaut.2024.103318.
11.
Yildirim P, Ceken C, Hassanpour R, Tolun MR. Prediction of similarities among rheumatic diseases. J Med Syst 2012; 36: 1485–1490, DOI: 10.1007/s10916-010-9609-6.
12.
Cafaro G, Bartoloni E, Baldini C, et al.; FIRMA (Interdisciplinary Forum for the Research on Autoimmune Diseases) Collaborators. Autoantibody status according to multiparametric assay accurately estimates connective tissue disease classification and identifies clinically relevant disease clusters. RMD Open 2023; 9: e003365, DOI: 10.1136/rmdopen-2023-003365.
13.
Vincent A, Hoskin TL, Whipple MO, et al. OMERACT-based fibromyalgia symptom subgroups: an exploratory cluster analysis. Arthritis Res Ther 2014; 16: 463, DOI: 10.1186/s13075-014-0463-7.
14.
Zhu H, Wu C, Jiang N, et al. Identification of 6 dermatomyositis subgroups using principal component analysis-based cluster analysis. Int J Rheum Dis 2019; 22: 1383–1392, DOI: 10.1111/1756-185X.13609.
15.
Zhang J, Xue Y, Liu X, et al. Identification of 4 subgroups in juvenile dermatomyositis by principal component analysis- based cluster analysis. Clin Exp Rheumatol 2022; 40: 443–449, DOI: 10.55563/clinexprheumatol/t2hxjd.
16.
Rider LG, Shah M, Mamyrova G, et al.; Childhood Myositis Heterogeneity Collaborative Study Group. The myositis autoantibody phenotypes of the juvenile idiopathic inflammatory myopathies. Medicine 2013; 92: 223–243, DOI: 10.1097/MD.0b013e31829d08f9.
17.
Li D, Tansley SL. Juvenile dermatomyositis-clinical phenotypes. Curr Rheumatol Rep 2019; 21: 74, DOI: 10.1007/s11926-019-0871-4.
18.
Sato S, Kuwana M. Clinically amyopathic dermatomyositis. Curr Opin Rheumatol 2010; 22: 639–643, DOI: 10.1097/BOR.0b013e32833f1987.
19.
Ye S, Chen XX, Lu XY, et al. Adult clinically amyopathic dermatomyositis with rapid progressive interstitial lung disease: a retrospective cohort study. Clin Rheumatol 2007; 26: 1647–1654, DOI: 10.1007/s10067-007-0562-9.
20.
Sato S, Uejima Y, Nanbu M, et al. Clinical analysis and outcome of interstitial lung disease complicated with juvenile dermatomyositis and juvenile polymyositis. Mod Rheumatol 2017; 27: 652–656, DOI: 10.1080/14397595.2016.1231102.
21.
Mukae H, Ishimoto H, Sakamoto N, et al. Clinical differences between interstitial lung disease associated with clinically amyopathic dermatomyositis and classic dermatomyositis. Chest 2009; 136: 1341–1347, DOI: 10.1378/chest.08-2740.
22.
Robinson AB, Reed AM. Clinical features, pathogenesis and treatment of juvenile and adult dermatomyositis. Nat Rev Rheumatol 2011; 7: 664–675, DOI: 10.1038/nrrheum.2011.139.
23.
Sabbagh S, Pinal-Fernandez I, Kishi T, et al. Anti-Ro52 autoantibodies are associated with interstitial lung disease and more severe disease in patients with juvenile myositis. Ann Rheum Dis 2019; 78: 988–995, DOI: 10.1136/annrheumdis- 2018-215004.
24.
Abu-Rumeileh S, Marrani E, Maniscalco V, et al. Lung involvement in juvenile idiopathic inflammatory myopathy: a systematic review. Autoimmun Rev 2023; 22: 103416, DOI: 10.1016/j.autrev.2023.103416.
25.
Kono H, Inokuma S, Nakayama H, Suzuki M. Pneumomediastinum in dermatomyositis: association with cutaneous vasculopathy. Ann Rheum Dis 2000; 59: 372–376, DOI: 10.1136/ard.59.5.372.
26.
Mahe E, Descamps V, Burnouf M, Crickx B. A helpful clinical sign predictive of cancer in adult dermatomyositis: cutaneous necrosis. Arch Dermatol 2003; 139: 539, DOI: 10.1001/archderm. 139.4.539-a.
27.
Narang NS, Casciola-Rosen L, Li S, et al. Cutaneous ulceration in dermatomyositis: association with anti-melanoma differentiation-associated gene 5 antibodies and interstitial lung disease. Arthritis Care Res (Hoboken) 2015; 67: 667–672, DOI: 10.1002/acr.22498.
28.
Barut K, Aydin P, Adrovic A, et al. Juvenile dermatomyositis: a tertiary center experience. Clin Rheumatol 2017; 36: 361–366, DOI: 10.1007/s10067-016-3530-4.
29.
Sag E, Demir S, Bilginer Y, et al. Clinical features, muscle biopsy scores, myositis specific antibody profiles and outcome in juvenile dermatomyositis. Semin Arthritis Rheum 2021; 51: 95–100, DOI: 10.1016/j.semarthrit.2020.10.007.
30.
Suzon B, Goulabchand R, Louis-Sidney F, et al. Subcutaneous tissue involvement in idiopathic inflammatory myopathies: Systematic literature review including three new cases and hypothetical mechanisms. Autoimmun Rev 2023; 22: 103284, DOI: 10.1016/j.autrev.2023.103284.
31.
McHugh NJ, Tansley SL. Autoantibodies in myositis. Nat Rev Rheumatol 2018; 14: 290–302, DOI: 10.1038/nrrheum.2018.56.
32.
Smith V, Pizzorni C, De Keyser F, et al. Reliability of the qualitative and semiquantitative nailfold videocapillaroscopy assessment in a systemic sclerosis cohort: a two-centre study. Ann Rheum Dis 2010; 69: 1092–1096, DOI: 10.1136/ard.2009. 115568.
33.
Sulli A, Secchi ME, Pizzorni C, Cutolo M. Scoring the nailfold microvascular changes during the capillaroscopic analysis in systemic sclerosis patients. Ann Rheum Dis 2008; 67: 885–887, DOI: 10.1136/ard.2007.079756. .
34.
van den Hoogen F, Khanna D, Fransen J, et al. 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis 2013; 72: 1747–1755, DOI: 10.1136/annrheumdis-2013-204424.
35.
Smith V, Riccieri V, Pizzorni C, et al. Nailfold capillaroscopy for prediction of novel future severe organ involvement in systemic sclerosis. J Rheumatol 2013; 40: 2023–2028, DOI: 10.3899/jrheum.130528.
36.
Riccieri V, Vasile M, Iannace N, et al. Systemic sclerosis patients with and without pulmonary arterial hypertension: a nailfold capillaroscopy study. Rheumatology 2013; 52: 1525–1528, DOI: 10.1093/rheumatology/ket168.
37.
Sulli A, Ruaro B, Smith V, et al. Progression of nailfold microvascular damage and antinuclear antibody pattern in systemic sclerosis. J Rheumatol 2013; 40: 634–639, DOI: 10.3899/jrheum.121089. .
38.
Cutolo M, Pizzorni C, Tuccio M, et al. Nailfold videocapillaroscopic patterns and serum autoantibodies in systemic sclerosis. Rheumatology 2004; 43: 719–726, DOI: 10.1093/ rheumatology/keh156.
39.
Sulli A, Pizzorni C, Smith V, et al. Timing of transition between capillaroscopic patterns in systemic sclerosis. Arthritis Rheum 2012; 64: 821–825, DOI: 10.1002/art.33463.
40.
Ganczarczyk ML, Lee P, Armstrong SK. Nailfold capillary microscopy in polymyositis and dermatomyositis. Arthritis Rheum 1988; 31: 116–119, DOI: 10.1002/art.1780310116.
41.
Selva-O’Callaghan A, Fonollosa-Pla V, Trallero-Araguas E, et al. Nailfold capillary microscopy in adults with inflammatory myopathy. Semin Arthritis Rheum 2010; 39: 398–404, DOI: 10.1016/j.semarthrit.2008.09.003.
42.
Mugii N, Hasegawa M, Matsushita T, et al. Association between nail-fold capillary findings and disease activity in dermatomyositis. Rheumatology 2011; 50: 1091–1098, DOI: 10.1093/rheumatology/keq430.
43.
Manfredi A, Sebastiani M, Cassone G, et al. Nailfold capillaroscopic changes in dermatomyositis and polymyositis. Clin Rheumatol 2015; 34: 279–284, DOI: 10.1007/s10067-014-2795-8.
44.
Piette Y, Reynaert V, Vanhaecke A, et al. Standardised interpretation of capillaroscopy in autoimmune idiopathic inflammatory myopathies: A structured review on behalf of the EULAR study group on microcirculation in Rheumatic Diseases. Autoimmun Rev 2022; 21: 103087, DOI: 10.1016/ j.autrev.2022.103087.
45.
Kubo S, Todoroki Y, Nakayamada S, et al. Significance of nailfold videocapillaroscopy in patients with idiopathic inflammatory myopathies. Rheumatology (Oxford) 2019; 58: 120–130, DOI: 10.1093/rheumatology/key257.
46.
Barth Z, Witczak BN, Flatø B, et al. Assessment of microvascular abnormalities by nailfold capillaroscopy in juvenile dermatomyositis after medium- to long-term followup. Arthritis Care Res 2018; 70: 768–776, DOI: 10.1002/acr.23338.
47.
Barth Z, Schwartz T, Flatø B, et al. Association between nailfold capillary density and pulmonary and cardiac ınvolvement in medium to longstanding juvenile dermatomyositis. Arthritis Care Res (Hoboken) 2019; 71: 492–497, DOI: 10.1002/acr.23687.
Copyright: © Narodowy Instytut Geriatrii, Reumatologii i Rehabilitacji w Warszawie. This is an Open Access journal, all articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International (CC BY-NC-SA 4.0) License (https://creativecommons.org/licenses/by-nc-sa/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, provided the original work is properly cited and states its license.
Udostępnij
ARTYKUŁ POWIĄZANY
Przetwarzamy dane osobowe zbierane podczas odwiedzania serwisu. Realizacja funkcji pozyskiwania informacji o użytkownikach i ich zachowaniu odbywa się poprzez dobrowolnie wprowadzone w formularzach informacje oraz zapisywanie w urządzeniach końcowych plików cookies (tzw. ciasteczka). Dane, w tym pliki cookies, wykorzystywane są w celu realizacji usług, zapewnienia wygodnego korzystania ze strony oraz w celu monitorowania ruchu zgodnie z Polityką prywatności. Dane są także zbierane i przetwarzane przez narzędzie Google Analytics (więcej).
Możesz zmienić ustawienia cookies w swojej przeglądarce. Ograniczenie stosowania plików cookies w konfiguracji przeglądarki może wpłynąć na niektóre funkcjonalności dostępne na stronie.
Możesz zmienić ustawienia cookies w swojej przeglądarce. Ograniczenie stosowania plików cookies w konfiguracji przeglądarki może wpłynąć na niektóre funkcjonalności dostępne na stronie.